Answers
Fission is the formation of two smaller atoms by the collision of the neutrons. The quantity of energy produced per gram is 7.23 × 10¹⁰ J/g.
What is nuclear fission?Nuclear fission is a process in which the neutron collides with the atom and results in the formation of two isotopes.
The nuclear fission reaction is shown as,
\(\rm ^{235}_{92}U + ^{1}_{0} n \rightarrow ^{144}_{54} Xe + ^{90}_{38}Sr + 2^{1}_{0}n\)
Energy in joules is calculated by the Einstein's formula as:
\(\begin{aligned} \rm E &= \rm mc^{2}\\\\&= 3.139 \times 10^{-28} \times (2.99 \times 10^{8})^{2}\\\\&= 2.82 \times 10^{-11}\;\rm J\end{aligned}\)
Energy per gram is calculated as:
\(\begin{aligned}& =\dfrac{ (2.82 \times 10^{-11})(6.022 \times 10^{26})}{235.043}\\\\&= 0.0722 \times 10^{12}\\\\&= 7.23 \times 10^{10} \;\rm J/g\end{aligned}\)
Therefore, the energy per gram of the fission is \(7.23 \times 10^{10} \;\rm J/g.\)
Learn more about fission here:
https://brainly.com/question/17137674
#SPJ1
Related Questions
Nitrogen-fixing bacteria convert atmospheric nitrogen into forms of nitrogen that living things can use.
Please select the best answer from the choices provided
•T
•F
Answers
Answer:
I know im late but its true
Explanation:
E2020 edge
Answer:
true
Explanation:
A fruit juice has a very sour taste. What does this most likely indicate?
A. The juice is a metal
B. The juice is a salt
C. The juice is an acid
D. The juice is a base
Answers
Explanation:
its hard its either B or C i dont wanna be wrong;-;
Answer:
The Juice wouldnt be a metal, Salt isnt that effective in juice if you have sensitive tastebuds. Your answer is C. THe Juice is an acid
Explanation:
Use this equation for the next question:
2NaOH + H2SO4 ® Na2SO4 + 2H20
If a reaction produces 0.75 moles Na2SO4, how many moles of NaOH were used?
0.75 moles NaOH
2 moles NaOH
.375 moles NaOH
1.5 moles NaOH
Answers
Calculate the mass of magnesium carbonate ( MgCO3), in grams, required to produce 110.0 g of carbon dioxide using the following equation: MgCO3 --> MgO CO2
Answers
Answer:
\(210.7~g~MgCO_3\)
Explanation:
We have to start with the reaction:
\(MgCO_3~->~MgO~+~CO_2\)
We have the same amount of atoms on both sides, so, we can continue. The next step is to find the number of moles that we have in the 110.0 g of carbon dioxide, to this, we have to know the atomic mass of each atom:
C: 12 g/mol
O: 16 g/mol
Mg: 23.3 g/mol
If we take into account the number of atoms in the formula, we can calculate the molar mass of carbon dioxide:
\((12*1)+(16*2)=44~g/mol\)
In other words: \(1~mol~CO_2=~44~g~CO_2\). With this in mind, we can calculate the moles:
\(110~g~CO_2\frac{1~mol~CO_2}{44~g~CO_2}=25~mol~CO_2\)
Now, the molar ratio between carbon dioxide and magnesium carbonate is 1:1, so:
\(2.5~mol~CO_2=2.5~mol~MgCO_3\)
With the molar mass of \(MgCO_3\) (\((23.3*1)+(12*1)+(16*3)=84.3~g/mol\). With this in mind, we can calculate the grams of magnesium carbonate:
\(2.5~mol~MgCO_3\frac{84.3~g~MgCO_3}{1~mol~MgCO_3}=210.7~g~MgCO_3\)
I hope it helps!
Give a reason for each of the following laboratory practices:
I. KMnO4 solution is not acidified with HNO3 before titration
II.FeSO4 solution is usually prepared fresh when needed
III. Alkali are not stored in glass stoped reagent bottles
Answers
Answer:
See explanation
Explanation:
The acid commonly used to acidify KMnO4 is H2SO4. The reason why HNO3 is not used is because HNO3 is itself a strong oxidizing agent. HCl is not used because it can react with KMnO4.
FeSO4 solution is usually prepared fresh when needed because if the solution is prepared and left to stand for sometime, FeSO4 is oxidized to Fe2(SO4)3 by air. Fe III does not give the brown ring test.
Alkali are not stored in glass stopped reagent bottles because very strong alkali can cause the stopper to corrode and fuse to the neck of the bottle making it very difficult to open the reagent bottle.
What would the expected temperature change be (in F) if 0.5 gram sample of water released 0.0501 j of heat energy? The specific heat of liquid water 4.184 j/g
Answers
The expected temperature change would be approximately 4.32°F if 0.5-gram sample of water released 0.0501 j of heat energy.
The specific heat of a substance is the amount of heat energy required to raise the temperature of one gram of the substance by one degree Celsius. The expected temperature change can be calculated using the formula:
q = m × c × ΔT
where q is the heat energy released, m is the mass of water, c is the specific heat of water, and ΔT is the change in temperature.
Rearranging the formula to solve for ΔT, we get:
ΔT = q ÷ (m × c)
Substituting the given values, we get:
ΔT = 0.0501 J ÷ (0.5 g × 4.184 J/g°C)
ΔT ≈ 2.4°C or 4.32°F
When a substance undergoes a change in temperature, it either gains or loses heat energy.
To learn more about temperature follow the link:
https://brainly.com/question/15267055
#SPJ1
Calcium carbonate is a common ingredient in antacids that reduces the discomfort associated with acidic stomach or heartburn. Stomach acid is hydrocholoric acid, HCl.
What volume in milliliters (mL) of an HCl
solution with a pH of 1.55
can be neutralized by 38.0 mg
of CaCO3?
Answers
The volume of acid we will require in this situation is 27000 mL.
Describe neutralization.We are aware that understanding the reaction equation is necessary because it is the first step in resolving the issue at hand;
Having said that,
CaCO3 + 2HCl → CaCl2 + H2O + CO2
Acid molarity = antilog (-1.55).
The number of base moles;
38 g/100 g/mol
= 0.38 moles
If 2 moles of acid and 1 mole of base combine;
The base responds with 0.38 * 2/1 moles.
= 0.76 moles
Volume = n/M
= 0.76/ 0.028
= 27 L or 27000 mL
Learn more about neutralization:brainly.com/question/14156911
#SPJ1
if two substance are at the same temperature, their enthalpy
Answers
Answer:
cannot be measure
Hope this helps :) !!!
The pOH of a solution is 6.0. Which statement is correct?
Use pOH = -log[OH-] and PH+pOH = 14.
The pH of the solution is 20.0.
O The concentration of OH ions is 1.0 x 108 M.
The concentration of OH ions is 1.0 x 106 M.
O The pH of the solution is 8.0.
A
Answers
At pOH value of 6.0 the pH value of the following solution is 8.0 and the concentration of [\(H^{+}\) ] ion is \(10^{-8}\)
In this question we will apply the formula
pH +pOH = 14 . . . . . . . . . . . . .(1)
where pH = concentration of [\(H^{+}\) ] ion
pOH = concentration of [\(OH^{-}\) ] ion
As per the question
pOH =6.0
Putting the value of pOH in equation (1) we get the value of pH
pH + 6.0 =14
pH = 14 -6.0
pH = 8.0
The value of pH if the pOH value is 6.0 is 8.0
To find the concentration of \(H^{+}\) ion we will use the following formula
This is calculated by the formula
[\(H^{+}\)} = \(10^{-pH}\)
where we will write the values of pH
Hence the concentration of [\(H^{+}\)} ion is \(10^{-8}\)
Therefore at pOH of 6.0 the pH value of the following solution is 8.0 and the concentration of [\(H^{+}\) ] ion is \(10^{-8}\)
Read more about pH
https://brainly.com/question/11300720
The complete question is -
What is the pH value and concentration of [\(H^{+}\) ] ion of the following if the pOH value of the solution is 6.0 ?
8.0g of certain gas occupies 5.6 L at STP.
A) How many moles of gas are present?
B) What is the molar mass of the gas?
C) What is the common atmospheric gas was collected?
Answers
Answer:
A) Using the ideal gas law, we can calculate the number of moles of gas present:
```
PV = nRT
```
where:
* P = pressure (atm) = 1 atm
* V = volume (L) = 5.6 L
* n = number of moles of gas
* R = ideal gas constant = 0.08206 L atm / mol K
* T = temperature (K) = 273.15 K
Solving for n, we get:
```
n = (P * V) / RT
```
```
n = (1 atm * 5.6 L) / (0.08206 L atm / mol K * 273.15 K)
```
```
n = 0.25 mol
```
Therefore, there are 0.25 moles of gas present.
B) The molar mass of the gas can be calculated by dividing the mass of the gas (8.0 g) by the number of moles of gas (0.25 mol):
```
Molar mass = Mass / n
```
```
Molar mass = 8.0 g / 0.25 mol
```
```
Molar mass = 32 g/mol
```
The molar mass of the gas is 32 g/mol.
C) The common atmospheric gas with a molar mass of 32 g/mol is oxygen (O2). Therefore, the gas that was collected is oxygen.
Explanation:
For the following reaction conditions select the correct statement regarding the reaction below.
OH Cl OTs OTs N TsCl W X Y Z N
a. W is the correct product.
b. X is the correct product.
c. Y is the correct product.
d. Z is the correct product since pyridine acts as a nucleophile.
e. Both X and Y are both formed in this reaction as a racemic mixture
Answers
Answer:
B
Explanation:
The appropriate diagram of the question is shown in the first image attached below.
From the diagram, we see the reaction of Cyclopentanol taking place under Tscl pyridine. We are to show the reaction mechanism and determine from the options, which appropriate product fits in.
So, from the reaction, the hydroxyl substituent reacts with Tscl where cl is being lost. This process is followed by an attack of N substituent on the pyridine with the Hydrogen atom and cleaves off for the structure to form a stable structure. The stereochemistry of the compound remains unchanged and it maintains its stick formula.
Thus, X is the appropriate and the correct product.


Is the following equation balanced? P + 3O₂ ----> 2P₂O₃
Answers
Answer:
4P+ 3(O2)= 2(P2O3)
Explanation:
Need to add 4 infront of P on left hand side to balance number of atoms on both sides
QUESTION 18 What is the volume in dm³ of 5.5 grams carbon dioxide (CO₂) at room temperature and pressure (r.t.p)? Given the atomic masses, C= 12 and O = 16
Answers
The volume of 5.5 grams of carbon dioxide (CO2) at room temperature and pressure is approximately 2.42 dm³.
To calculate the volume of carbon dioxide (CO2) at room temperature and pressure (r.t.p), we can use the ideal gas law equation:
PV = nRT
Where:
P is the pressure
V is the volume
n is the number of moles
R is the ideal gas constant
T is the temperature
At r.t.p, the pressure is typically considered to be 1 atmosphere (atm) or 101.325 kilopascals (kPa). The temperature is approximately 298 Kelvin (K), which is equivalent to 25 degrees Celsius (°C).
First, we need to calculate the number of moles of carbon dioxide (CO2) using its mass and molar mass.
The molar mass of CO2 is calculated by adding the atomic masses of carbon (C) and two oxygen (O) atoms:
Molar mass of CO2 = (12.01 g/mol) + 2*(16.00 g/mol) = 44.01 g/mol
Now, we can calculate the number of moles using the given mass of 5.5 grams:
n = mass / molar mass = 5.5 g / 44.01 g/mol ≈ 0.125 mol
Next, we can rearrange the ideal gas law equation to solve for volume (V):
V = (nRT) / P
Substituting the known values into the equation:
V = (0.125 mol * 0.0821 L·atm/mol·K * 298 K) / 1 atm
V ≈ 2.42 dm³
for more such questions on volume
https://brainly.com/question/29796637
#SPJ11
Which one of the following is a main-group nonmetal element in period number 3 of the periodic table? sodium gallium bromine selenium none of these sulfur
Answers
Sulphur is a main-group nonmetal element in period number 3 of the periodic table.
Elements can be classified as metals, non-metals, or metalloids. Metals are excellent conductors of heat and electricity, and are malleable (can be swaged into sheets) and ductile (can be stretched into wires). Most metals are solid at room temperature and have a characteristic silvery luster (except mercury, which is liquid). Nonmetals are (usually) poor thermal and electrical conductors and are not malleable or ductile. Many of the elemental nonmetals are gases at room temperature, others are liquids, and others are solids. Semimetals are intermediate in their properties. Their physical properties are similar to non-metals, but some can be made conductive under certain circumstances. These semiconductors are very important in computers and other electronic devices.
Learn more about Non-metals here:
https://brainly.com/question/27696417
#SPj4
Distillation is a method that depends on differences in the boiling points of liquids to separate the components of a liquid mixture. Explain why you would separate a homogeneous mixture of two miscible liquids in this way but not a mixture of two immiscible liquids.
Answers
Explanation:
for immiscible liquids it is quite easy to separate and the separating funnel can be used but for miscible liquid they form a single entity and separating them is quite impossible if the differences in temperature is not considered,so in distillation the one with lower boiling point evaporates out living behind the one with high boiling point
I need help only with 2 questions please help me out thank you.The atom represented by the formula I is:IronIndiumPotassium Iodine
Answers
We can look for the symbol in the period table. Looking for it we see that is in group VII. It has an atomic number equal to 53 and its name is Iodine.
Solid calcium chlorate decomposes to form solid calcium chloride and oxygen gas.
Write the balanced chemical equation for the reaction described. Phases are optional.
equation:
Answers
Answer:
Ca(ClO₃)₂(s) → CaCl₂(s) + 3O₂(g)
Explanation:
Chemical equation:
Ca(ClO₃)₂(s) → CaCl₂(s) + O₂(g)
Balance chemical equation:
Ca(ClO₃)₂(s) → CaCl₂(s) + 3O₂(g)
Step 1:
Ca(ClO₃)₂(s) → CaCl₂(s) + O₂(g)
Left hand side Right hand side
Ca = 1 Ca = 1
Cl = 2 Cl = 2
O = 6 O = 2
Step 2:
Ca(ClO₃)₂(s) → CaCl₂(s) + 3O₂(g)
Left hand side Right hand side
Ca = 1 Ca = 1
Cl = 2 Cl = 2
O = 6 O = 6
HELLLP MEEEE
Write the chemical formula for each of the given compounds.

Answers
Answer:
sodium perchorate is NaCLO4
calcium sulfite CaSO3
potassium hydroxide KOH
lithium nitrate LiNO3
Explanation:
sodium perchorate has one Sodium
one Chlorine and 4 Oxygen
calcium sulfite has one Calcium on sulfate and 3Oxygen
lithium nitrate has one Lithium oneNitrogen
3 Oxygen
Solution Notes
8. Calculate the molarity of 500 ml of 0.0300 moles of NaOH.
Answers
Answer:
\(\huge\boxed{\sf M = 0.06\ M}\)
Explanation:
Given data:No. of moles = n = 0.03 mol
Volume = v = 500 ml = 0.5 L
Required:Molarity = M = ?
Formula:M = n / v
Solution:Put the given data in the above formula.
M = 0.03 / 0.5
M = 0.06 M\(\rule[225]{225}{2}\)
After considering the results of the classification tests, the possibilities can be narrowed down further by determining the melting point of the
A derivative.
B Schiff addition product.
C iodoform.
D triiodo compound.
Answers
After considering the results of the classification tests, the possibilities can be narrowed down further by determining the melting point of the B Schiff addition product.
What two facts can you infer about your product based on its melting point range?
A compound's melting point is helpful in two ways: it provides information on the composition's identity and degree of purity. Impurities will affect a compound's melting point, resulting in broader and lower melting point ranges.
First, the solubility test must be performed in order to remove soluble contaminants from the target molecule. The desired chemical and the soluble impurities are dissolved in a minimum of near or boiling solvent once a suitable solvent has been selected. The solution is then given time to gradually cool.
Learn more about Schiff addition product refer
https://brainly.com/question/14748028
#SPJ4
what winds that push the surface of a body of water
Answers
Answer:
i dont know if i understand real good
Answer:
upwelling
Explanation:
upwelling is the process of winds blowing across the ocean surface to push water away.
another aldol reaction could have occurred in the same reaction mixture. the enolate of acetone could have added to another molecule of acetone [instead of adding to a molecule of benzaldehyde]. predict the structure of the product if the acetone undergoes an aldol addition and dehydration with another molecule of acetone
Answers
Acetone enolate preferentially attack the benzaldehyde rather than another acetone molecule because it is a strong nucleophile which will attack the carbonyl of benzaldehyde and forms a B- carbonyl alkoxide ion.
Benzalacetone is the end product of the aldol condensation reaction between acetone and benzaldehyde. Acetone may be deprotonated to produce a nucleophilic enolate anion since it contains -hydrogens (on both sides). Because it is significantly more electrophilic than a ketone, the aldehyde carbonyl interacts with the enolate quickly. Since it dehydrates spontaneously under the reaction conditions, the original aldol adduct cannot be separated. However, the resulting, - α, β-unsaturated ketone contains active hydrogen that can condense with additional benzaldehyde molecules.
To learn more about Benzalacetone visit:https://brainly.com/question/19340462
#SPJ4
if u trust urself do it
Study these images.
4 photos of clouds. 1: Sky covered with large, flat layers of blue, grey clouds. 2: A tall, fluffy cloud shaped like an anvil. 3: Round, puffy clouds in a blue sky. 4: Thin, wispy clouds high in the sky.
Which image shows a cumulonimbus cloud?
1
2
3

Answers
Answer:
3
Explanation:
I wish you the best, its three or 2
Answer:
2
Explanation:
Edge 2021
Which one violates the octet rule?
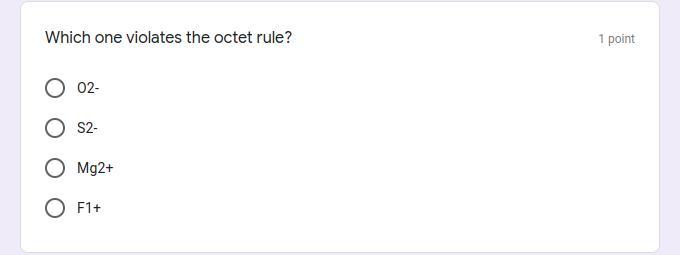
Answers
Answer:
F1+ is the one that violates the octet rule
Explanation:
F1+ does not end up having 8 electrons in its valence shell, and there for violates the octet rule, for an atom to comply with the octet rule its charge must reflect the number of electrons it needs to gain or lose to make an octet, but F1+'s charge does not reflect the number of electrons it needs to be stable or have an electron configuration of a noble gas
Relations to my budget

Answers
When there is an increase in an activity, like sales or manufacturing, the overall amount of an expense, known as a fixed expense, does not change.
Thus, Normal definitions typically include the phrase within a relevant or appropriate range of activity since a change is likely to take place at fixed expense either an exceptionally high or low volume or expense.
Of course, the rent will probably need to adjust if sales quadruple or fall to 20% of the average level. However, as the extreme circumstances are outside of the relevant range for short-term analysis, the current rent of $2,000 is regarded as a fixed expense.)
The following are some instances of costs that are probably set within a fair range of retail sales, The yearly pay for the shop manager.
Thus, When there is an increase in an activity, like sales or manufacturing, the overall amount of an expense, known as a fixed expense, does not change.
Learn more about Expenses, refer to the link:
https://brainly.com/question/29850561
#SPJ1
What is Heat? What is Cold?
Answers
Answer:
Heat is the process of something getting hot(temperatures going up). Cold is when something gets cold(temperature decreases).
Explanation:
Antimony reacts with sulfur to form Sb2S3.
What is the percentage yield for the reaction in which 1.40 g of Sb2S3 is obtained from 1.73 g of antimony and a slight excess of sulfur?
Answers
Answer:
.2sb
Explanation:
according to my mind its the percentage of the question
Which change to an object would double its kinetic energy?
A. Increasing its velocity to twice its original value
B. Increasing its mass to four times its original value
C. Increasing its mass to twice its original value
Ο Ο
D. Increasing its velocity to four times its original value
Answers
Answer:
Increasing its mass to twice its original value
Explanation:
Pls mark me brainliest
Many gyms and health clubs have steam saunas, which are small steam-filled rooms. Traditionally, steam saunas have a container of heated rocks. A small ladle of water is poured on the rocks in order to make the steam. Use what you have learned so far about heat transfer to explain how hot rocks can be used to make steam.
Answers
Solution :
It is given that a now-a-days many of the health clubs and the gyms provides steam saunas with the help of heated rocks in a container. When water is poured in to these heated rocks, steam is being produce.
This is because the energy conversion takes place in this process. The hot rocks have high temperatures and possess heat energy in them. So when cold water is poured in to the rocks, the cold water absorbs the heat energy from the rocks and is converted in to hot vapor by converting heat energy in to vapor energy or steam energy by the process of vaporization.
What is the balanced net ionic equation when aqueous solutions of Pb(NO3)2 and KBr are mixed and form a product
Answers
Answer:
KBr+Pb(NO3)2=? is an equation you can use.
Explanation:
