Answers
The name for H2SO4(aq) is Sulfuric acid.
Sulfuric acid (H2SO4) is a highly corrosive and strong acid. It is one of the most important industrial chemicals produced worldwide. Sulfuric acid is commonly used in various industries for a wide range of applications. It is used in the production of fertilizers, detergents, dyes, pharmaceuticals, and various chemical processes.
Sulfuric acid is also utilized in laboratories for its acidic properties and as a dehydrating agent due to its strong affinity for water. It is a dense, oily liquid that is colorless when pure but can appear yellowish or brownish due to impurities. When handling sulfuric acid, caution must be exercised as it can cause severe burns and is harmful if ingested or inhaled.
Learn more about Sulfuric acid on:
https://brainly.com/question/1107054
#SPJ1
Related Questions
Write a balanced equation for the combustion of gaseous propane (C3H8) a minority component of natural gas in which it combines with gaseous oxygen to form gaseous carbon dioxide
Answers
Answer:
C3H8 + 3O2 -------------> 3CO2 + 4H2 (Balanced Reaction)
13a. [3 marks]
A 4.406 g sample of a compound containing only C, H and O was burnt in excess oxygen. 8.802 g of CO2 and 3.604 g of H20 were produced.
Determine the empirical formula of the compound.
Answers
The empirical formula of the original compound given the combustion analysis data is C₂H₄O
How to determine the mass of Carbon Mass of CO₂ = 8.802 gMolar mass of CO₂ = 44 g/mol Molar of C = 12 g/mol Mass of C =?Mass of C = (12 / 44) × 8.802
Mass of C = 2.4 g
How to determine the mass of HMass of H₂O = 3.604 gMolar mass of H₂O = 18 g/mol Molar of H = 2 × 1 = 2 g/mol Mass of H =?Mass of H = (2 / 18) × 3.604
Mass of H = 0.4 g
How to determine the mass of O
Mass of compound = 4.406 gMass of C = 2.4 gMass of H = 0.4 gMass of O =?Mass of O = (mass of compound) – (mass of C + mass of H)
Mass of O = 4.406 – (2.4 + 0.4)
Mass of O = 1.606 g
How to determine the empirical formula C = 2.4 gH = 0.4 gO = 1.606 gEmpirical formula =?Divide by their molar mass
C = 2.4 / 12 = 0.2
H = 0.4 / 1 = 0.4
O = 1.606 / 16 = 0.1
Divide by the smallest
C = 0.2 / 0.1 = 2
H = 0.4 / 0.1 = 4
O = 0.1 / 0.1 = 1
Thus, the empirical formula of the compound is C₂H₄O
Learn more about empirical formula:
https://brainly.com/question/9459553
#SPJ1
3.
The color of the powdered form of a mineral is called
O fracture
O cleavage
O luster
O streak
Answers
Answer:
D. Streak.
Explanation:
A mineral, in simple terms, can be defined as a solid inorganic substance that occurs in a pure form with a specific crystal structure. A mineral is identified based on its physical properties which include luster, color, streak, crystal shape, etc.
The color of the powdered form of a mineral is called a streak. A streak is a useful property in distinguishing sulfide and oxide minerals. To check the color of a mineral, it is rubbed against the surface of a ceramic streak plate or a piece of unglazed ceramic. In its finely powdered form, a mineral is easy to identify.
Therefore, option D is correct.
the second (si unit of time) is defined as the duration of 9192631770 cycles of the radiation corresponding to the transition between levels of the fundamental state of the cesium-133 atom.
a. true
b. false
Answers
A second is defined as the time taken by 9192631770 radiation cycles to shift the cesium-133 atom's fundamental state from one level to another (si unit of time).
Simply expressed, the study of the chemical compound that radiation, typically ionizing radiation, induces in matter is known as radiation chemistry. Common themes in the field include radiation-induced particle nucleation and growth, radiation-enhanced H 2 generation, A second is defined as the time taken by 9192631770 radiation cycles to shift the cesium-133 atom's fundamental state. And damaging effects on nuclear system chemistry. Due to its ground state electron state's [Xe] 6s1 configuration, the caesium atom's atomic term symbol is 2 S 1/2. This shows that the atom has a single unpaired electron and an overall electron spin of 1/2.
Learn more about chemical compound here
https://brainly.com/question/12166462
#SPJ4
Work can be produced by passing the vapor phase of a two-phase substance stored in a tank through a turbine as shown in the given figure. Consider such a system using R-134a, which is initially at 80°F, and a 16-ft3 tank that initially is entirely filled with liquid R-134a. The turbine is isentropic, the temperature in the storage tank remains constant as mass is removed from it, and the R-134a leaves the turbine at 10 psia. How much work will be produced when the half of the liquid mass in the tank is used? Use data from tables.
Answers
The initial enthalpy, specific volume, and entropy are determined from the temperature and the state from A-11E:
h1 = 38.17 Btu/lbm
sf = 0.07934 Btu/lbm-R
vf =0.01334 ft3/lbm
The enthalpy at the outlet of the turbine is determined from the enthalpies of the constituents at the final pressure and the quality is expressed through the entropies were taken from A-12E:
=(3.153+((0.7934-0.00741)/0.22208)*95.536) Btu/lbm
=34.1 Btu/lbm
The energy balance determines work output:
mh1 = W + mh2
W = m(h1 - h2)
= V/2vf (h1 - h2)
=16/2*0.01334(38.17-34.1) Btu
= 2440 Btu
Therefore, The work that will be produced when the half of the liquid mass in the tank is used is 2440 Btu.
Hence 2440 Btu is a correct answer.
To know more about entropy follow link
https://brainly.com/question/13135498
#SPJ9
How many grams do 4.5 moles of oxygen atoms weigh
Answers
Answer:
71.99100
Explanation:
The mass of oxygen equal to one mole of oxygen is 15.998 grams and the mass of one mole of hydrogen is 1.008 g. 15.998*4.5 = 71.99100
Which of the following is NOT true of zinc?
-Excess zinc can decrease copper absorption.
-Grains are the most reliable food sources of zinc.
-All of its functions involve it acting as a cofactor for enzymes.
-It binds to most proteins in the body.
Answers
while zinc is an important mineral with numerous functions in the body, it is not true that grains are the most reliable food source of zinc. A balanced diet that includes a variety of foods can provide adequate zinc intake for most people.
How to solve the problem?
The statement that is NOT true of zinc is "Grains are the most reliable food sources of zinc." While grains can be a source of zinc, they are not necessarily the most reliable source.
Zinc is an essential mineral that plays important roles in many biological processes, including immune function, protein synthesis, wound healing, and DNA synthesis. It is involved in various enzymatic reactions, and acts as a cofactor for many enzymes. Zinc is also important for proper growth and development, especially during childhood and adolescence.
Excess zinc intake can lead to decreased copper absorption, as both minerals compete for absorption in the intestines. This can lead to copper deficiency, which can cause anemia neutropenia, and other health problems. Therefore, it is important to maintain a balance between zinc and copper intake.
While grains can be a source of zinc, other foods such as meat, seafood, and dairy products are also good sources. Vegetarians and vegans may need to pay particular attention to their zinc intake, as plant-based sources of zinc may be less bioavailable than -based sources. Zinc supplements can also be used to prevent or treat deficiencies, but should be used with caution as excessive intake can have negative health effects.
In summary, while zinc is an important mineral with numerous functions in the body, it is not true that grains are the most reliable food source of zinc. A balanced diet that includes a variety of foods can provide adequate zinc intake for most people.
To know more about enzymes visit :-
https://brainly.com/question/14577353
#SPJ1
Draw two resonance forms for nitrosyl chloride, ONCl. Which one is better and why?
Answers
Explanation:
this molecule does not exhibit any resonance, and even if it does, this structure would be the most viable since the net charge on all of the atoms are 0.

4. Long answer type questions: a. b. C. d. e. f. g. h. j. i. What are the constituent gases of air? Why is the surrounding air not seen with the eyes? How do you prove that air supports burning? How do you show that air occupies space? How do you prove that air has weight? How is air useful to us? Mention any three points. Write any three properties of air. How can you say that air exerts force? Write any four effects of air pollution. Write any three causes of air pollution and any two control measures of it.
Answers
1. The constituent gases of air are:
Nitrogen Oxygen Argon Carbon Dioxide2. The surrounding air is not seen with the eyes because it is transparent. Air molecules are not visible to the na-ked eye, and they do not scatter or absorb visible light significantly. Therefore, air appears colorless and transparent.
What is air?3. To prove that air supports burning, you can perform an experiment with a burning candle. Place a glass jar or bell jar over a lit candle, ensuring that the jar is airtight. As the candle burns, it consumes oxygen from the air inside the jar. Eventually, the candle flame will go out due to the lack of oxygen, proving that air (specifically oxygen) is necessary for burning.
4. To show that air occupies space, you can perform a simple experiment using a plastic bottle or syringe. Fill the bottle or syringe with water, ensuring there are no air bubbles. Then, cover the opening tightly and try to compress the air inside. You will find that it is not possible to compress the air significantly, indicating that air occupies space.
5. To prove that air has weight, you can use a sensitive balance or scale. Weigh an airtight container or balloon, and then fill it with air. The weight of the container or balloon with the added air will be greater than its initial weight, demonstrating that air has weight.
6. Air is useful to us in various ways. Three points highlighting the importance of air are:
Breathing and RespirationCombustion and Energy ProductionClimate Regulation7. Three properties of air include:
Air is Compressible: Air can be compressed or expanded under different conditions, allowing it to fill various spaces and containers.Air has Mass: Air molecules have mass, which means air itself has weight. It exerts pressure on objects and surfaces.Air Exerts Pressure: Due to the collisions of air molecules with surfaces, air exerts pressure in all directions. This pressure is known as atmospheric pressure.Air exerts force in various ways. For example, air pressure allows objects like airplanes to fly by providing lift. Air resistance or drag opposes the motion of objects moving through the air, creating a force that can affect their speed and trajectory.
8. Four effects of air pollution include:
Respiratory ProblemsEnvironmental Damage:Climate ChangeHuman Health Impacts9. Causes of pollution:
Industrial EmissionsVehicle EmissionsResidential and Agricultural Activities10. Two control measures for air pollution include:
Emission ReductionAir Quality RegulationsLearn more about air on https://brainly.com/question/15215203
#SPJ1
mixing sand and gravel and knitting yarn into a scarf
Answers
Mixing sand and gravel and knitting yarn into a scarf are examples of physical changes.
What are physical changes?Physical changes are defined as changes which affect only the form of a substance but not it's chemical composition. They are used to separate mixtures in to chemical components but cannot be used to separate compounds to simpler compounds.
Physical changes are always reversible using physical means and involve a change in the physical properties.Examples of physical changes include melting,boiling , change in texture, size,color,volume and density.Magnetism, crystallization, formation of alloys are all reversible and hence physical changes.
Learn more about physical changes,here:
https://brainly.com/question/13216903
#SPJ1
Your question is incomplete, but most probably your full question was mixing sand and gravel and knitting yarn into a scarf is a physical change or chemical change.
The sun is 93 million miles from the earth approximately how many years would it take to travel from earth to the sun if a person flew in a spaceship at 55 miles an hour?
(Hint:begin by dividing the distance by the speed to determine the number of hours it would take. Round your answers the the nearest whole number.)
Answers
Answer:
it would take 2 years
Explanation:
93/55=1.69--->1.70------>2
Question 1 (2 points)
What is the mass in mg of 9.3 x 1016 lead atoms? Give answer with correct
significant figures and units.
Answers
Answer:
the answer would be 9,448.8
The mass of 9.3 × 10¹⁶ lead atoms is equal to 0.032 mg.
What is Avogadro's number?Avogadro’s number represents the number of entities in one mole of any substance. Generally, these units can be molecules, atoms, ions, electrons, or protons, depending upon the type of chemical reaction or reactant and product.
The value of Avogadro’s constant is approximately equal to 6.022 × 10²³ mol⁻¹.
Given, the number of atoms of the lead = 9.3 × 10¹⁶
The atomic mass of the lead = 207.2 g/mol
So 6.022 × 10²³ atoms of lead have mass = 207.2g
Then 9.3 × 10¹⁶ lead atoms will have mass \(=\frac{207.2\times 9.3\times 10^{23}}{ 6.022\times 10^{23}}\) = 31.998 ×10⁻⁷g
As we know, 1 gram = 1000 mg
The mass of the 9.3 × 10¹⁶ lead atoms = 31.998 ×10⁻⁴ mg = 0.0032 mg
Therefore, the mass of 9.3 × 10¹⁶ lead atoms is 0.032 mg.
Learn more about Avogadro's number, here:
brainly.com/question/11907018
#SPJ2
a) At 40 °C, what the saturated solubility level of KCl (aq)? (1 mark)
b) If 40 g of KCl is added to 100 g of water at 90 °C, would the solution be saturated? Explain. (2 marks)
c) Describe two ways in which saturation can be achieved if only 40 g of KCl is added to 100 g of water at 90 ° (3 marks)

Answers
a) The saturated solubility level of KCl (aq) at 40°C is 35.7 g/100 mL of water.
b) No, the solution would not be saturated at 90°C. At this temperature, the solubility of KCl in water is higher than at 40°C. Therefore, more KCl can dissolve in 100 g of water at 90°C than at 40°C. To determine if the solution is saturated, we need to compare the amount of KCl that actually dissolved in the water to the maximum amount of KCl that can dissolve in the water at that temperature. If the amount of KCl that dissolved is less than the maximum amount, then the solution is unsaturated. If the amount of KCl that dissolved is equal to the maximum amount, then the solution is saturated. If the amount of KCl that dissolved is greater than the maximum amount, then the solution is supersaturated.
c) Two ways in which saturation can be achieved if only 40 g of KCl is added to 100 g of water at 90°C are:
i) Heating the solution to a higher temperature: As the temperature of the solution is increased, the solubility of KCl in water also increases. Therefore, by heating the solution to a higher temperature than 90°C, more KCl can be dissolved in the water until the solution becomes saturated.
ii) Allowing the solution to cool slowly: If the solutionis heated to a temperature higher than 90°C and then allowed to cool slowly, the solubility of KCl in water decreases as the temperature decreases. This means that as the solution cools, KCl will begin to precipitate out of the solution until the solution becomes saturated. Alternatively, if the solution is left undisturbed at 90°C and allowed to cool slowly, KCl will begin to precipitate out of the solution as it reaches its saturation point.
Balance the following reaction by typing in the correct coefficients in front of each reactant and product.
H3PO4(s) -
-->
H₂(g) +
P(s) +
O₂(g)
Answers
Consider the cell Pt |Cr²+ (aq, 1.0 M), Cr3+ (aq, 2.2 mM) || Pb2+ (aq, 1.3M)| Pb. EºCell -0.37. What is the value of K at 25 °C
Answers
Answer:
1
Explanation:
To determine the value of K (equilibrium constant) at 25 °C, we can use the Nernst equation, which relates the cell potential (E) to the equilibrium constant (K) and the standard cell potential (EºCell). The Nernst equation is given by:
E = EºCell - (RT / nF) * ln(K)
Where:
E = cell potential
EºCell = standard cell potential
R = gas constant (8.314 J/(mol·K))
T = temperature in Kelvin (25 °C = 298 K)
n = number of electrons transferred in the balanced redox equation
F = Faraday's constant (96,485 C/mol)
ln = natural logarithm
In this case, the given standard cell potential (EºCell) is -0.37 V.
The balanced redox equation for the cell reaction is:
Pt + Cr²+ -> Pt + Cr³+
Since there is no change in the oxidation state of Pt, no electrons are transferred in the reaction (n = 0).
Substituting the known values into the Nernst equation, we have:
E = -0.37 V - (8.314 J/(mol·K) * 298 K / (0 * 96,485 C/mol)) * ln(K)
E = -0.37 V
Since n = 0, the term (RT / nF) * ln(K) becomes 0, and we are left with:
-0.37 V = -0.37 V - 0
This implies that the value of K is 1, since any number raised to the power of 0 is equal to 1.
Therefore, the value of K at 25 °C for the given cell is 1.
Question
Arrange the elements according to atomic radius.
Largest radius to Smallest radius
Answer Bank
K
Ca
Ga
Ge
As
Sc
Br
Kr

Answers
K
Ca
Ga
Ge
As
Br
Kr
Smallest
The elements according to the decreasing atomic radius are arranged as-
K, Ca, Sc, Ga, Ge, As, Br, Kr
An atomic radius is half the distance between adjacent atoms of the same element in a molecule. It is a measure of the size of the element’s atoms, which is typically the mean distance from the nucleus centre to the boundary of its surrounding shells of the electrons.
An atom gets larger as the number of electronic shells increase; therefore the radius of atoms increases as you go down a certain group in the periodic table of elements. The atomic radius decreases on moving from left to right across a period.
Thus the elements according to the decreasing atomic radius are arranged as -
K, Ca, Sc, Ga, Ge, As, Br, Kr
Learn more about Atomic radius, here:
https://brainly.com/question/13963619
#SPJ6
How many moles of O2 are dissolved n 10.0 L of a 0.001 mol dm-3 solution?
Answers
Answer:
\(0.01molO_2\)
Explanation:
Hello there!
In this case, by considering the definition of molar solution as the moles of solute over volume in liters or cubic meters of solution, we can see that actually 10.0 L of the solution are equal to 10.0 dm³. In such a way, since the concentration is 0.001 mol / dm³, the moles of oxygen that are dissolved turn out to be:
\(0.001mol/dm^3 * 10.0dm^3\\\\0.01molO_2\)
Best regards!
can someone help me with my chemistry homework please???

Answers
1.) Lithium and Sulfide:
Formula: \(\bold{Li_{2}S}\)Ion Charges: \(\bold{Li~1+,~Li~1+,~S~2-}\)2.) Lithium and Chlorine:
Formula: \(\bold{2LiCl}\)Ion Charges: \(\bold{Li~1+, Li~1+,Cl~1-,Cl~1-}\)3.) Lithium and Oxygen:
Formula: \(\bold{Li_{2}O}\)Ion Charges: \(\bold{Li~1+,Li~1+,O~2-}\)4.) Lithium and Nitrogen:
Formula: \(\bold{Li_{3}N}\)Ion Charges: \(\bold{Li~1+,Li~1+,Li~1+,N~3-}\)5.) Magnesium and Sulfur:
Formula: \(\bold{MgS}\)Ion Charges: \(\bold{Mg~2+,S~2-}\)6.) Magnesium and Chlorine:
Formula: \(\bold{MgCl_2}\)Ion Charges: \(\bold{Mg~2+,Cl~1-,Cl~1-}\)7.) Magnesium and Oxygen:
Formula: \(\bold{MgO}\)Ion Charges: \(\bold{Mg~2+,O~2-}\)8.) Magnesium and Nitrogen:
Formula: \(\bold{Mg_3N_2}\)Ion Charges: \(\bold{Mg~2+,Mg~2+,Mg~2+,N~3-,N~3-}\)Explanation:______________________________
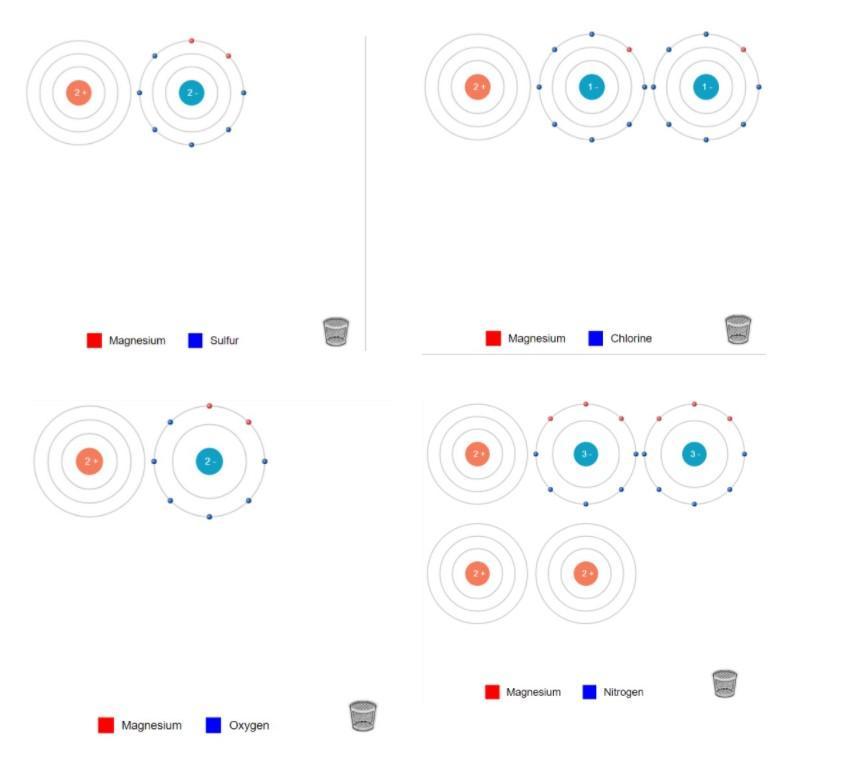
Lithium and Sulfur: In order to make Lithium Sulfide, There must be 2 Lithium and 1 Sulfur. You transfer the electrons from both Lithium's to the Sulfur.Lithium and Chlorine:In order to make Lithium Chloride, There must be 2 Lithium and 2 Chlorine. You transfer the electrons from both Lithium's to the Chlorines, (One electron for each chlorine.)Lithium and Oxygen:In order to make Lithium Oxide, There must be 2 Lithium and 1 Oxygen. You transfer the electrons from both Lithium to Oxygen. Lithium and Nitrogen:In order to make Lithium Nitride, There must be 3 Lithium and 1 Nitrogen. You transfer the electrons from all 3 Lithium to Nitrogen. Magnesium and Sulfur:In order to make Magnesium Sulfide, There must be 1 Magnesium and 1 Sulfur. You transfer the both electrons from Magnesium to Sulfur. Magnesium and Chlorine:In order to make Magnesium Chloride, There must be 1 Magnesium and 2 Chlorine. You transfer on electron to each Chlorine. Magnesium and Oxygen:In order to make Magnesium Oxide, There must be 1 Magnesium and 1 Oxygen. You transfer both electrons from Magnesium to Oxygen. Magnesium and Nitrogen:In order to make Magnesium Nitride, There must be 3 Magnesium and 2 Nitrogen. You transfer 3 electrons from Magnesium to Nitrogen.______________________________

PLS HELP THE QUESTION IS ON THE PICTURE

Answers
Concepts used:
1 mole of an element or a compound has 6.022 * 10²³ formula units
So, we can say that: Number of formula units = number of moles * 6.022*10²³
number of moles of an element or a compound = given mass/molar mass
__________________________________________________________
003 - Number of CaH₂ formula units in 6.065 grams
Number of Moles:
We know that the molar mass of CaH₂ is 42 grams/mol
Number of Moles of CaH₂ = given mass/molar mass
Number of moles = 6.065 / 42
Number of moles = 0.143 moles
Number of Formula units:
Number of formula units = number of moles * 6.022*10²³
= 0.143 * 6.022 * 10²³
= 0.86 * 10²³ formula units
__________________________________________________________
004 - Mass of 6.34 * 10²⁴ formula units of NaBF₄
Number of Moles:
We mentioned this formula before:
Number of formula units = number of moles * 6.022*10²³
Solving it for number of moles, we get:
Number of moles = Number of Formula units / 6.022* 10²³
replacing the variable
Number of moles = 6.34 * 10²⁴ / 6.022*10²³
Number of moles= 10.5 moles
Mass of 10.5 moles of NaBF₄:
Molar mass of NaBF₄ = 38 grams/mol
Mass of 10.5 moles = 10.5 * molar mass
Mass of 10.5 moles = 10.5 * 38
Mass = 399 grams
__________________________________________________________
005 - Number of moles in 9.78 * 10²¹ formula units of CeI₃
Number of Moles:
We have the formula:
Number of moles = Number of Formula units / 6.022* 10²³
replacing the variables
Number of Moles = 9.78 * 10²¹ / 6.022*10²³
Number of Moles = 1.6 / 10²
Number of Moles = 1.6 * 10⁻² moles OR 0.016 moles
What is the Internal Energy of a Human Body? Define internal energy as Delta E.
SHOW ALL WORK!
20 points
Answers
The internal energy of a human body is the net energy contained in the body due motion of its particle or molecules.
In a human body the internal energy is stored . It increases when the temperature of the body rises, or when the body observes some changes. Internal energy we can say that is the sum of kinetic and potential energy of all particles in the body.
Internal energy is a state function of a system and is an extensive quantity. Every substance possesses a fixed quantity of energy which depends upon its chemical nature and also on its state of existence. Every substance have a definite value of Internal energy.
The change in the Internal energy of a reaction may be considered as the difference between the internal energies of the two states.
ΔU = \(E_{B}\) - \(E_{A}\) where \(E_{B}\) and \(E_{A}\) are the initial energies of states A and B. Or we can also write the equation as:
ΔU = ΔE
To know more about Internal energy
https://brainly.com/question/13125947
#SPJ1
convert 650 mi to mm
Answers
Answer:
1046073600 millimeters
Which of the following best identifies where long-range order would be found?
in amorphous solids
in crystalline solids
in thermal plasmas
in nonthermal plasmas
Answers
Long-range order is best found in crystalline solids. Crystalline solids are characterized by a highly ordered arrangement of atoms or molecules in a repeating pattern called a crystal lattice. Option 2)
Long-range order is best found in crystalline solids. Crystalline solids are characterized by a highly ordered arrangement of atoms or molecules in a repeating pattern called a crystal lattice. The arrangement of the particles in a crystalline solid extends over long distances, resulting in a well-defined and repetitive structure.
In contrast, amorphous solids lack long-range order. They do not possess a well-defined crystal lattice or a regular arrangement of particles. The atoms or molecules in amorphous solids are arranged in a more random and disordered manner, leading to a lack of long-range order.
Thermal plasmas and nonthermal plasmas, on the other hand, are states of matter where the atoms or molecules are highly ionized, resulting in the formation of charged particles or ions. While plasmas can exhibit certain levels of order, they do not possess the same level of long-range order as crystalline solids.
Therefore, crystalline solids are the primary location where long-range order is typically found. Option 2)
For more question on atoms
https://brainly.com/question/6258301
#SPJ8
Note Complete Question
Which of the following best identifies where long-range order would be found?
1) in amorphous solids
2) in crystalline solids
3) in thermal plasmas
4) in nonthermal plasmas
OPTION B. Long-range order is most commonly found in crystalline solids, where atoms or molecules are arranged in a well-defined, consistent pattern. Amorphous solids and plasmas do not showcase this characteristic.
Explanation:Long-range order pertains to the organization and regularity of constituents over large scale distances. It's a term often associated with the structure of crystalline solids. This is due to the fact that their atoms or molecules are arranged in a well-defined and repeated pattern extending in all three spatial dimensions over the length of the solid. On the contrary, an amorphous solid does not have a long-range order, instead, its structure is only ordered on a short range. Plasmas, both thermal or nonthermal, are an ionized gas and do not exhibit long-range order, primarily due to their high level of kinetic energy and lack of fixed positions for particles.
Learn more about Long-Range Order here:https://brainly.com/question/10474914
#SPJ12
describe the dual nature of electrons (give an example)
Answers
Answer:
In 1924, the French physicist, Louis de Broglie suggested that if light has electron, behaves both as a material particle and as a wave. According to this theory, small particles like electrons when in motion possess wave properties.
Explanation:
examples
This can be derived as follows according to Planck’s equation, E = hv = hc /λ ∴ v=c/λ(energy of the photon (on the basis of Einstein’s mass energy relationship) E = mc2
( If Bohr’s theory is associated with de-Broglie’s equation then wave length of an electron can be determined in bohr’s orbit and relate it with circumference and multiply with a whole number 2πr = nλ or λ = (2πr/2π) From de-Broglie equation, λ = (h/mv).
The equilibrium constant for the following reaction:
Ca(HCO3)2(s) <--> CaO(s) + 2 CO2(g) + H2O(g)
would be:
Group of answer choices
A. K = [Ca(HCO3)2] / [CaO][CO2]2[H2O]
B. K = [CaO][CO2]2[H2O] / [Ca(HCO3)2]
C. K = [CO2]2[H2O]
D. K = [CO2]2[H2O] / [Ca(HCO3)2]
Answers
Answer:
C.) \(K = {[CO_{2} ]^{2} [H_2O] }\)
Explanation:
The general equilibrium expression looks like this:
\(K = \frac{[B]^b[C]^{c} [D]^{d} }{[A]^{a}} }\)
In this expression,
-----> K = equilibrium constant
-----> uppercase letters = formulas
-----> lowercase letters = balanced equation coefficients
Solids are not included in the expression, so Ca(HCO₃)₂(s) and CaO(s) should be left out. The products should be in the numerator and the reactants are in the denominator. This makes the expression:
\(K = \frac{[B]^b[C]^{c} [D]^{d} }{[A]^{a}} }\) <----- Equilibrium expression
\(K = {[CO_{2} ]^{c} [H_2O]^{d} }\) <----- Insert gaseous formulas
\(K = {[CO_{2} ]^{2} [H_2O] }\) <----- Insert coefficients
A solution of aluminum chloride has a pH of (4.5x10^0). What is the [H3O*(aq)], in mol/L?
Note: Your answer is assumed to be reduced to the highest power possible.
Answers
The concentration of H3O+ ions in the solution of aluminum chloride is \(3.16×10^-5\) mol/L.
Aluminum chloride is an acidic salt that contains a cation, Al3+, and an anion, Cl-. When aluminum chloride is dissolved in water, it dissociates into its constituent ions, and the Al3+ cations hydrolyze to produce H+ ions.
This reaction leads to the formation of an acidic solution. The pH of a solution of aluminum chloride is \(4.5×10^0\). We need to determine the concentration of H3O+ ions in this solution.
The concentration of H3O+ ions in a solution is given by the equation: pH = -log[H3O+] where pH is the negative logarithm of the concentration of H3O+ ions in the solution. The negative sign indicates that the pH is inversely proportional to the concentration of H3O+ ions. To determine the concentration of H3O+ ions, we need to rearrange the equation:
[H3O+] = \(10^-pH\) Substituting the value of pH =\(4.5×10^0\), we get: [H3O+] = \(10^-4.5\)
The value of \(10^-4.5\) can be calculated using scientific notation: \(10^-4.5\)= \(3.16×10^-5\) mol/L
for more such questions on concentration
https://brainly.com/question/28564792
#SPJ8
A 488.3 gram sample of an unknown substance (MM = 92.41 g/mol) is heated from -23.1 °C to 51.8 °C. (heat capacity of solid = 2.96 J/g・°C; heat capacity of liquid = 1.75 J/g・°C; ∆Hfus = 8.04 kJ/mol; Tfinal = 17.6 °C)
a) How much energy (in kJ) is absorbed/released to heat the solid?
b)How much energy (in kJ) is absorbed/released to melt the solid?
c)How much energy (in kJ) is absorbed/released to heat the liquid?
d) What is the total amount of energy that must be absorbed/released for the entire process?
Answers
Answer:
a) Q₁ = 58.82 KJ
b) Q₂ = 42.48 KJ
c) Q₃ = 29.22 KJ
d) Q = 130.52 KJ
Explanation:
a)
In order to find the energy absorbed to heat the solid, we will use:
\(Q_{1} = mC_{1}\Delta T_{1}\)
where,
Q₁ = Heat absorbed for heating solid = ?
m = mass of solid = 488.3 g = 0.4883 kg
C₁ = Specific Heat Capacity of Solid = 2.96 J/g °C
ΔT₁ = Change in temperature of Solid = Melting Temperature - Initial Temp.
ΔT₁ = 17.6°C - (-23.1°C) = 40.7°C
Therefore,
\(Q_{1} = (488.3\ g)(2.96\ J/g\ ^{0}C)(40.7\ ^{0}C)\)
Q₁ = 58.82 KJ
b)
In order to find the absorbed to melt the solid at 17.6°C, we will use:
\(Q_{2} = nH_{fus}\)
where,
Q₂ = Heat absorbed for melting solid = ?
H_fus = Heat of Fusion = 8.04 KJ/mol
n = no. of moles = \(\frac{m}{MM} = \frac{488.3\ g}{92.41\ g/mol} = 5.28 mol\)
Therefore,
\(Q_{2} = (5.28\ mol)(8.04\ KJ/mol)\)
Q₂ = 42.48 KJ
c)
In order to find the energy absorbed to heat the liquid, we will use:
\(Q_{3} = m C_{3}\Delta T_{3}\)
where,
Q₃ = Heat absorbed for heating Liquid = ?
m = mass of solid = 488.3 g = 0.4883 kg
C₃ = Specific Heat Capacity of Liquid = 1.75 J/g °C
ΔT₃ = Change in temperature of Liquid = Final Temp. - Melting Temp.
ΔT₃ = 51.8°C - 17.6°C = 34.2°C
Therefore,
\(Q_{3} = (488.3\ g)(1.75\ J/g\ ^{0}C)(34.2\ ^{0}C)\)
Q₃ = 29.22 KJ
d)
Total amount of energy absorbed during entire process is:
\(Q = Q_{1} + Q_{2} + Q_{3}\)
\(Q = 58.82\ KJ + 42.48\ KJ + 29.22\ KJ\)
Q = 130.52 KJ
In order to heat a 488.3 g solid, 58.8 kJ are required. To melt the solid, 42.5 kJ are required. To heat the liquid, 29.2 kJ are required. The total amount of energy absorbed is 130.5 kJ.
Initially, a 488.3 g solid at -23.1 °C is heated up to 17.6 °C (melting point). We can calculate the heat required (Q₁) using the following expression.
\(Q_1 = c \times m \times \Delta T = \frac{2.96J}{g.\° C } \times 488.3g \times (17.6\° C-(-23.1\° C)) \times \frac{1kJ}{1000J} = 58.8 kJ\)
where,
c: heat capacity of the solidm: massΔT: change in the temperatureAt 17.6 °C, we can calculate the heat (Q₂) required to melt the solid using the following expression.
\(Q_2 = \Delta H_{fus} \times \frac{m}{MM} = 8.04 kJ/mol \times \frac{488.3 g}{92.41g/mol} = 42.5kJ\)
where,
∆Hfus: enthalpy of fusionm: massMM: molar massThe liquid is heated from 17.6 °C to 51.8 °C. We can calculate the heat required (Q₃) using the following expression.
\(Q_3 = c \times m \times \Delta T = \frac{1.75J}{g.\° C } \times 488.3g \times (51.8\° C-17.6\° C)) \times \frac{1kJ}{1000J} = 29.2 kJ\)
c: heat capacity of the liquidm: massΔT: change in the temperatureThe total amount of energy absorbed (Q) is the sum of the energy absorbed in each step.
\(Q = Q_1 + Q_2 + Q_3 = 58.8kJ+42.5kJ+29.2kJ= 130.5kJ\)
In order to heat a 488.3 g solid, 58.8 kJ are required. To melt the solid, 42.5 kJ are required. To heat the liquid, 29.2 kJ are required. The total amount of energy absorbed is 130.5 kJ.
Learn more: https://brainly.com/question/10481356

What do you think would happen to the Earth’s energy balance if the concentration of greenhouse gases increase
Answers
Answer:
rise to an imbalance in Earth's energy budget
Explanation:
Increased atmospheric greenhouse gas concentrations give rise to an imbalance in Earth's energy budget by initially reducing the amount of emitted thermal infra-red radiation. The result of this imbalance is an accumulation of excess energy in the Earth system over time.
Answer:
Increased atmospheric greenhouse gas concentrations give rise to an imbalance in Earth's energy budget by initially reducing the amount of emitted thermal infra-red radiation. The result of this imbalance is an accumulation of excess energy in the Earth system over time.
Explanation:
hope it helps you
1) To increase the amount of NH3 at 200 atm, the manufacturer should (increase, decrease, not change) the temperature of the reaction chamber.
2) This change in temperature would shift the reaction to the (left, right) because this equilibrium reaction is (exothermic, endothermic)
Answers
The temperature of the reaction should be decreased
This change in temperature would make the equilibrium to shift to the right.
What is the LeChatelier principle?
The Le Chatelier's principle, commonly referred to as the Le Chatelier's principle of equilibrium, is a chemical principle that describes how an equilibrium system reacts to environmental changes.
According to this theory, when an equilibrium system is exposed to an outside force, it will respond in a way that partially offsets the imposed change and restore equilibrium.
Learn more about LeChatelier principle:https://brainly.com/question/31377984
#SPJ1
The weak base ammonia, NH3, and the strong acid hydrobromic acid react to form the salt ammonium bromide, NH4Br. Given that the value of Kb for ammonia is 1.8×10−5, what is the pH of a 0.053 M solution of ammonium bromide at 25∘C?
Answers
The pH of a 0.053 M solution of ammonium bromide at 25° C is equal to 33.9.
What are acids and bases?Acids are a substance that has a pH lower than 7 and bases are a substance that has a pH of more than 7.
the pH of any solution will be calculated as:
pH = -log[H⁺]
Given the chemical reaction in equilibrium with ICE table will be:
NH₄⁺ + H₂O ⇌ NH₃ + H₃O⁺
Initial: 0.289 0 0
Change: -x +x +x
Equilibrium: 0.289-x x x
Equation for Kb for this reaction is:
Kb = [NH₃][H₃O⁺] / [NH₄⁺]
Given value of Kb = 1.8 × 10⁻⁵
On putting values equation becomes
1.8 × 10⁻⁵ = x² / 0.053 - x
The value of x is negligible as compared to 0.053 so the equation becomes
1.8 × 10⁻⁵ = x² / 0.053
x = 2.28 × 10⁻³
i.e. [NH₃] = [H₃O⁺] = 33.9 × 10⁻³
Calculating the pH by putting this concentration value on the equation
pH = -log(33.9 × 10⁻³)
pH = 33.9
Thus, the pH will be 33.9.
To learn more about acids and bases, refer to the link:
https://brainly.com/question/13646534
#SPJ1
Vinegar is sold at the grocery store with a concentration of 5.0 % acetic acid. How many grams of acetic acid are in 28 g of Vinegar?
Answers
White vinegar typically consists of 93%–96% water and 4–7% acetic acid. It can be used to cooking, bake, cleaning, and get rid of weeds. It can also help you lose weight and lower your blood sugar and cholesterol. Consumption is safe in moderation, but excessive consumption or when combined with certain medications could be harmful.
Apple cider vinegar is widely used in cooking and as a salad dressing because it contains acetic acid and nutrients like vitamins C and B vitamins. But at the same time, it's been utilized customarily as medication. It helps in losing weight.
Learn more about vinegar, here:
https://brainly.com/question/23700611
#SPJ1