Answers
Answer:
Explanation:
These are daily things u use
Related Questions
For a concave mirror, incident light beams through C will reflect:
(a) back through C
(b) through F
(c) parallel to PA
Answers
Answer:
c
Explanation:
HELP ME PLEASE !!!
One time I went to the mountains. I was scared of altitude sickness, so I got cannisters of oxgen (O₂). These cannisters contain 2L of compressed oxygen (O₂). When the oxygen is pressurized, it condenses into its liquid form inside the cannister. How much would this oxygen (0₂) weigh in its liquid form?
2.9 grams
1.6 grams
3.6 grams
4.3 grams
Answers
Answer:2.9 grams
Explanation:i had this question
2.
Which mixture could be a useful buffer in a solution?
acetic acid (CH3CO2H) and hydrochloric acid (HCl)
sodium hydroxide (NaOH) and elemental sodium (Na)
ammonia (NH3) and ammonium chloride (NH4Cl)
acetic acid (CH3CO2H) and ammonia (NH3)
Pls answer quickly
Answers
Ammonia (\(NH_3\)) and ammonium chloride (\(NH_4Cl\)) mixture could be a useful buffer in a solution. Option C
A buffer is a solution that can resist changes in pH when small amounts of acid or base are added. It consists of a weak acid and its conjugate base or a weak base and its conjugate acid. The buffer system works by the principle of Le Chatelier's principle, where the equilibrium is shifted to counteract the changes caused by the addition of an acid or a base.
In option A, acetic acid (\(CH_3CO_2H\)) is a weak acid, but hydrochloric acid (HCl) is a strong acid. This combination does not form a buffer because HCl is completely dissociated in water and cannot provide a significant concentration of its conjugate base.
Option B consists of sodium hydroxide (NaOH), which is a strong base, and elemental sodium (Na), which is a metal. This combination does not form a buffer as there is no weak acid-base pair involved.
Option D contains acetic acid (\(CH_3CO_2H\)), a weak acid, and ammonia (\(NH_3\)), a weak base. Although they are weak acid and base, they do not form a buffer system together as they are both weak acids or bases and lack the required conjugate acid-base pair.
Option C, ammonia (\(NH_3\)), is a weak base, and ammonium chloride (\(NH_4Cl\)) is its conjugate acid. This combination can form a buffer system. When ammonia reacts with water, it forms ammonium ions (NH4+) and hydroxide ions (OH-).
The ammonium ions act as the weak acid, while the ammonia acts as the weak base. The addition of a small amount of acid will be counteracted by the ammonium ions, and the addition of a small amount of base will be counteracted by the ammonia, thus maintaining the pH of the solution relatively stable.
Therefore, option C, consisting of ammonia (\(NH_3\)) and ammonium chloride (\(NH_4Cl\)), is the suitable mixture that could be a useful buffer in a solution.
For more such question on buffer visit:
https://brainly.com/question/13076037
#SPJ8
Part B: What happens to the speed of Mercury as moves closer to the Sun in its orbit?
It speeds up
It slows down
Its speed remains constant throughout its orbit
Answers
Answer:
It speeds around the Sun every 88 days, traveling through space at nearly 29 miles (47 kilometers) per second, faster than any other planet. Mercury spins slowly on its axis and completes one rotation every 59 Earth days.
When you say that
something is hard to lift, are you talking about
weight or mass?
Answers
Answer:
Weight
Explanation:
WHen talking about weight you’re referring to the force exerted to lift if, but when you’re talking about mass it’s just how much it weighs but not how much it takes to lift it.
What is the part of an experiment that is the measurable outcome?
Answers
Answer: - your answer should be the Dependent Variable
Hope that helps
9. Which of the following gas laws is calculated with the pressure and
volume variables at a constant temperature?
Formula
4 points
P₁V₁ = P₂V₂
P₁ = first pressure
P2 = second pressure
V₁ = first volume
Answers
The gas law that is calculated with the pressure and volume variables at a constant temperature is Boyle's Law. Boyle's Law states that the pressure (P) of a gas is inversely proportional to its volume (V) when temperature (T) is held constant.
Mathematically, it is expressed as P₁V₁ = P₂V₂, where P₁ and V₁ represent the initial pressure and volume, and P₂ and V₂ represent the final pressure and volume.According to Boyle's Law, if the volume of a gas is reduced while keeping the temperature constant, the pressure will increase proportionally.
Similarly, if the volume is increased, the pressure will decrease. This relationship holds as long as the temperature remains constant throughout the process. Boyle's Law is one of the fundamental gas laws and provides insights into the behavior of gases under changing pressure and volume conditions at a constant temperature.
For more such questions on gas law
https://brainly.com/question/30233942
#SPJ11
PLS HELP ME WITH THIS ILL GIVE BRAINLIEST

Answers
Answer:
1 qualiative physical
2 qualiative chemical
3 quantitative physcial
4 quantiative chemical
5qualiative physcial
6quatiative chemcial
7quanatiative physcial
8 qualiative chemical
9quatliate physcial
A quantity of nitrogen gas is enclosed in a tightly stoppered 500 mL flask at room temperature (20.0 °C) and 0.836 atm pressure. The flask is then heated to 680 °C. If the flask can withstand pressures up to 3 atm, will it explode under this heating?
Answers
The vessel will not explode since the new gas pressure is less than 3 atm.
What will be the pressure when the temperature of a given volume of nitrogen gas is heated?The pressure when the temperature of a given volume of nitrogen gas is heated is calculated using the formula given below:
P₁/T₁ = P₂/T₂where;
P₁ = initial pressureT₁ = initial temperatureP₂= final pressureT₂ = final temperatureSolving for P₂
P₂ = P₁ * T₂ / T₁
P₂ = 0.836 * (680 + 273) / (20 + 273)
P₂ = 2.72 atm
The new pressure is less than 3 atm.
Learn more about gas pressure at: https://brainly.com/question/25736513
#SPJ1
Calculate the percent composition (out of 100) by weight of potassium in potassium carbonate decahydrate.
Answers
Answer:
12.26%
Explanation:
MM K = 39.1 g/mol
MM K2CO3 . 10H2O = 2x39.1 + 12.01 + 3 x 15.99 + 10 x 18.02= 318.38 g/mol
% by weight = (MM K/MM K2CO3.10H2O ) x 100= (39.1/318.8) x 100=12.26 %
The percent composition by weight of potassium in potassium carbonate decahydrate is 13.98%
Let's represent the chemical formula of the compound as follows;
potassium carbonate decahydrate = CH₂0KO₁₃
The molecular mass of the compound can be calculated as follows:
Molecular mass of CH₂₀KO₁₃molecular mass = 12 + 20 + 39 + (16×13) = 279 g/molmass of potassium = 39 g
Therefore,
% mass of potassium = 39 / 279 × 100
% mass of potassium = 3900 / 279
% mass of potassium = 13.9784946237
% mass of potassium = 13.98 %
learn more on percentage composition here: https://brainly.com/question/3905173
Which of the following would be considered a strong base?
a H₂SO4
b. CO₂
c. Ca(OH)₂
d. C₁₂H₂O..
Answers
It’s name is calcium hydroxide and often it is easy to tell if something is a strong base if it has (OH) in the name.
What is the amount of charge on a calcium ion if its neutral atom has lost two valence electrons?
A. 1+
B. 2+
C. 3+
D. 4+
Answers
Answer: B
Explanation: a calcium ion has a charge of +2 because it has 2 more protons than electrons giving it a positive charge instead of neutral or negative.
Hope it will help you
Determine the pH at the equivalence point in the titration of 50.0 ml of 0.300 m ch3cooh with .3 M NaOH. the Value of Ka for CH3COOH is 1.8 x 10^-5
Answers
The pH at the equivalence point in the titration of 50.0 ml of 0.300 M CH₃COOH with 0.3 M NaOH is 8.61.
At the equivalence point in the titration of CH₃COOH with NaOH, the pH is determined by the hydrolysis of the salt formed, which is CH3COO⁻ and Na+.
The equation for the hydrolysis reaction is:
CH3COO⁻ + H2O ⇌ CH3COOH + OH⁻
We can use the Ka value for CH₃COOH to find the Kb value for CH3COO⁻ using the equation:
Kw = Ka x Kb
Where Kw is the ion product constant for water (1.0 x 10⁻¹⁴).
Kb = Kw/Ka
= (1.0 x 10⁻¹⁴)/(1.8 x 10⁻⁵)
= 5.56 x 10⁻¹⁰
Now we can use the Kb value to find the concentration of OH⁻ at the equivalence point using the equation:
Kb = ([OH⁻][CH3COOH])/[CH3COO⁻].
Since the concentrations of CH3COOH and CH3COO⁻ are equal at the equivalence point, we can simplify the equation to Kb = ([OH⁻]²)/[CH₃COO⁻].
Hence,
[OH-] = \(\sqrt{Kb(CH3COO-)}\)
=\(\sqrt{5.56(10^{-10})(0.3) }\)
= 4.06 x 10⁻⁶ M
Use the concentration of OH⁻ to find the pH at the equivalence point using the equation pOH = -log[OH-] and the relationship pH + pOH = 14.
pOH = -log(4.06 x 10⁻⁶)
= 5.39
pH = 14 - 5.39
= 8.61
Therefore, the pH at the equivalence point in the titration of 50.0 ml of 0.300 M CH3COOH with 0.3 M NaOH is 8.61.
Learn more about pH here: https://brainly.com/question/172153
#SPJ11
A drawing.Short description, A drawing.,Long description,
The drawing shows a wave of water moving to the right in a container. As the wave moves, the distance between the peak and trough of the wave gets shorter, but the distance between the peaks of the waves remains the same.
Question
What does the drawing show about the energy of the wave as it moves?
Answer options with 4 options
A.
The energy of the wave increases as shown by the increasing frequency.
B.
The energy of the wave decreases as shown by the decreasing amplitude.
C.
The energy of the wave remains the same as shown by the same wavelength.
D.
The energy of the wave regularly increases and decreases as shown by the peaks and troughs.
the picture is this:

Answers
Answer:
wow 15 points is alot
Explanation:
Why does an increased temperature cause a reaction to occur slower? A. The increased temperature makes the molecules more resistant to sucessful collision, they bounce off of each other more often. B. The increased kinetic energy causes the particles to move faster, causing more collisions. C. It does not. The increased temperature causes the reaction to occur more quickly. D. The increased potential energy in the particles means more energy is needed from the environment for the activation energy.
Answers
An increased temperature generally causes a reaction to occur faster rather than slower. Therefore option C is correct.
The increased temperature leads to a higher average kinetic energy of the molecules, which results in more frequent and energetic collisions between the reactant particles.
This increased collision frequency and energy facilitate the breaking of chemical bonds and the formation of new bonds, leading to an accelerated reaction rate.
When the temperature is raised, the kinetic energy of the molecules increases. This means that the individual molecules move faster and possess a greater amount of energy. As a result, the molecules collide more frequently and with higher energy, enhancing the likelihood of successful collisions that lead to a reaction.
In summary, an increased temperature leads to a faster reaction rate by increasing the average kinetic energy of the particles, causing more frequent and energetic collisions.
To know more about temperature-
brainly.com/question/15520591
#SPJ1
An infant acetaminophen suspension contains 80.0mg/0.80 mL suspension. The recommended dose is 15 mg/kg body weight. (1.000 lb. is equivalent to 453.59 g; this is a measured equality.)
How many mL of this suspension should be given to an infant weighing 17 lb ? (Assume two significant figures.)
Express your answer using two significant figures.
Answers
The amount, in mL, of the suspension that should be given to an infant weighing 17 lb will be 1.16 mL
Dimensional analysis0.8 mL of the liquid contains 80.0 mg of the drug.
The recommended dose is 15 mg per kg of body weight
The infant to be given the drug weighs 17 lb.
First, let's convert the weight of the infant to kg.
1 lb = 453.59 g
17 lb = 453.59 x 17/1
= 7711.03 g
1000 g = 1 kg
7711.03 g = 7711.03 x 1/1000
= 7.711 kg
So, the baby's weight is 7.711 kg.
The drug dose for the baby can thus be calculated as:
15 mg x 7.711 = 115.67 mg
But 0.8 mL of the drug contains only 80.0 mg. How many mL will contain 115.67 mg?
0.8 x 115.67/ 80.0 = 1.16 mL
More on dimensional analysis can be found here: https://brainly.com/question/13078117
#SPJ1
The city council of a small town wanted to add fluoride to their water so that all the residents would have healthier teeth. The mayor had read that ionic compounds must be used to get the fluoride into water. He looked up fluorine-containing compounds and came up with the following list.
NaF NF3 SiF4 CaF2 NH4F
Using academic vocabulary in paragraph form, discuss the following:
Explain which compound(s) could produce fluoride ions in the water. In your response, be sure to include:
° which of the listed compounds are ionic and which are covalent.
° how ionic and covalent compounds are different.
° why only ionic compounds would produce fluoride in water.
Answers
Answer:NaF is ionic. NF3 is covalent. SiF4 is ionic. CaF2 is Ionic and NH4F is also ionic. Ionic compounds transfer electrons whereas covalent compounds share electrons hence the word "co". Also, ionic compounds are formed with metal and nonmetal. Where a covalent is with 2 nonmetals. Only ionic compounds would produce fluoride in water because ionic compounds can dissolve in water and covalent compounds cant.
The compounds that can be used for the release of fluorine in water are NaF, \(\rm SiF_4\), \(\rm CaF_2\), and \(\rm NH_4F\).
The ionic compounds are formed by the transfer of electrons between the atoms of the compounds. The ionic bonds are weaker and are easy to dissociate in the aqueous solution.
The covalent compounds are formed by the sharing of electrons between the atoms. Since there is sharing, there has been a strong bond, that has been hard to dissociate.
The Mayor has to release Fluoride in the water, thus ionic compounds are preferred that have the easy release of fluorine.
From the given compounds:
NaF = Ionic compound\(\rm NF_3\) = Covalent compound\(\rm SiF_4\) = Ionic compound\(\rm CaF_2\) = Ionic compound\(\rm NH_4F\) = Ionic compoundThus the compounds that can be used for the release of fluorine in water are NaF, \(\rm SiF_4\), \(\rm CaF_2\), and \(\rm NH_4F\).
For more information about ionic and covalent compounds, refer to the link:
https://brainly.com/question/10748806
Where Did the Water
in the Puddle Go
Six friends were walking to the park on a sunny day. They noticed a big puddle
on their way to the park. When they came back two hours later, the puddle was
gone. They each had different ideas about where the water in the puddle went.
This is what they said:
Bandari: The water went right up to the clouds.
Regina: The sun dried the water up and it no longer exists.
Dylan: All of the water soaked into the ground.
Axel: Much of the water is in the air around us.
The water went into a stream, river, pond, lake, or ocean.
Clinti: The water changed into fog.
Explain your
Which friend do you think has the best idea?
thinking
Answers
Answer:
Axel: Much of the water is in the air around us.
Explanation:
When a puddle of water forms on the ground, it indicates that much of the ground around it is saturated with water and as such, the water would not readily soak into the ground. Also, since the puddle was found in a park, it is not likely that the water has gone into a stream, river, pond, lake, or ocean.
Since, the day was a sunny, it means that the heat from the sun has caused the evaporation of the water molecules. However, the water vapor molecules do not just immediately rise up to the atmosphere to form clouds nor do they cease to exist, rather it is dispersed in the air around the surroundings and beyond. Formation of clouds by water vapor takes days to happen.
Also, the water has not changed to fog either as they form usually at cooler temperatures.
I NEED HELP PLS:(
10 mL of 1 M HBr at 20 °C is mixed with 25 mL of 1 M NaOH at 20 °C in a calorimeter. The temperature increases to 23°C.How much heat was released? The density of water is 1g/mL. The specific heat of water is 4.18 J/g˚C.
Answers
The heat released is 439.5 J during the reaction.
The heat released can be calculated using the formula:
q = mCΔTwhere q is the heat released, m is the mass of the solution, C is the specific heat of water, and ΔT is the change in temperature.
First, we need to calculate the total mass of the solution:
10 mL HBr + 25 mL NaOH = 35 mLThe density of water is 1 g/mL, so the mass of the solution is 35 g.
Next, we can calculate the change in temperature:
ΔT = final temperature - initial temperatureΔT = 23°C - 20°CΔT = 3°CFinally, we can substitute these values into the formula:
q = 35 g * 4.18 J/g°C * 3°Cq = 439.5 JTherefore, 439.5 J of heat was released during the reaction.
To learn more about calorimeter, here
https://brainly.com/question/4802333
#SPJ1
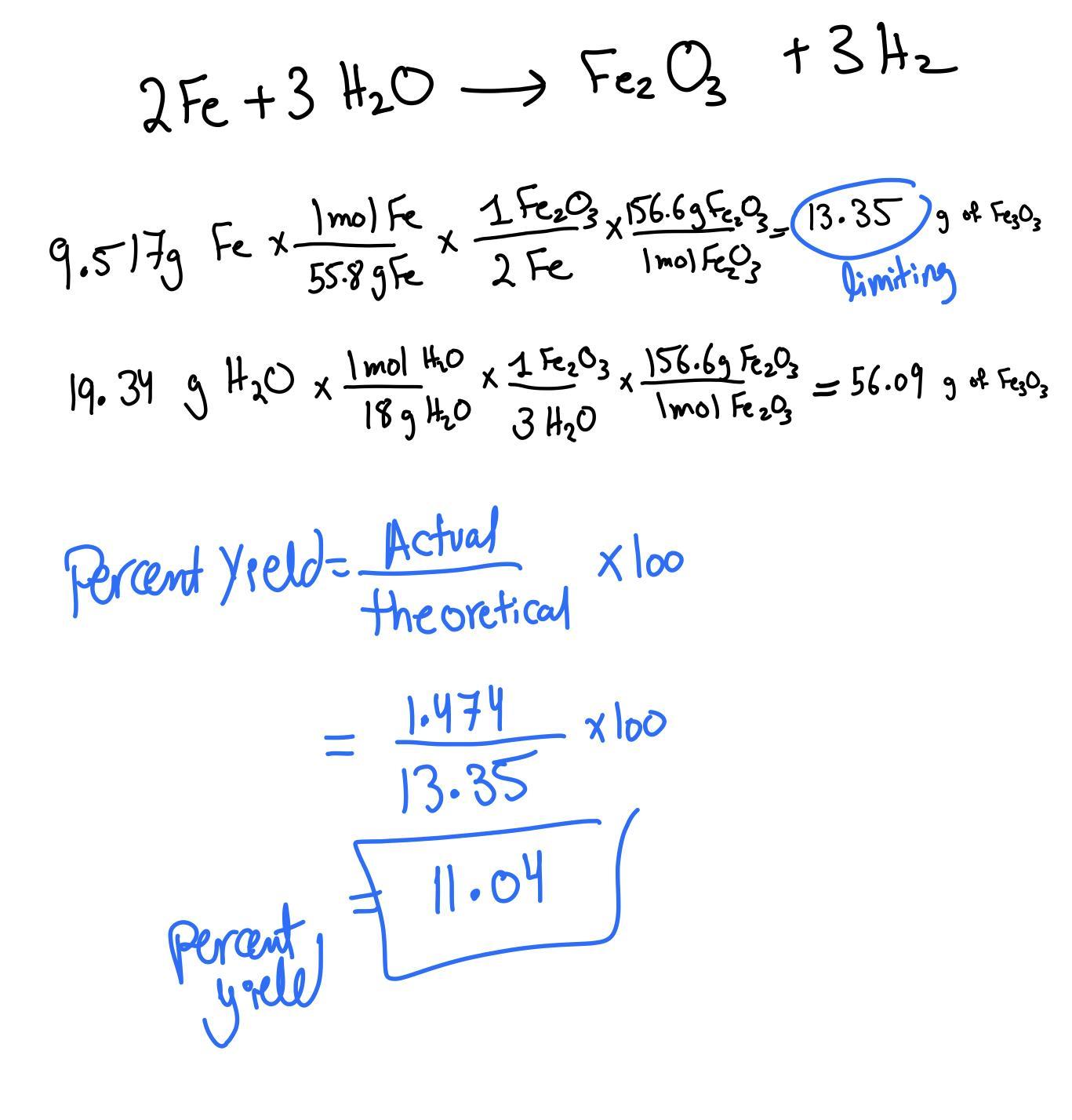
oud sand lime teWhat is the limiting reagent when 9.517 g of Fe is allowed to react with 19.34 g of water according to
the reaction given below? Base your calculations on the yield of Fe2O3.
2 Fe + 3 H20 -› Fe203 + 3 H2
Answers
Answer:
Theoretical Yield = 13.35 g of Fe2O3
Explanation:
Check the conversion to get to the limiting reaction below!
A new element was discovered. It’s average atomic mass was found to be 245.6 amu. Isotope #1 has a mass of 243.2 amu; isotope #2 has a mass of 244.9 amu; isotope #3 has a mass of 247.2 amu. Which isotope is most likely to be the most abundant?
A
Isotope 1
B
Isotope 2
C
Isotope 3
D
They all contribute the same amount
Answers
Answer:
B. Isotope 2
Explanation:
Isotopes can be defined as the atom of an element that has the same number of protons but different number of neutrons. This ultimately implies that, the isotopes of an element have the same atomic number (number of protons) but different atomic mass (number of nucleons).
Basically, the atomic mass of any chemical element found in the periodic table is equal to the weighted average of all its isotopes. Therefore, the isotope with the closest mass to the weighted average would be the most abundant.
In this scenario, a new element was discovered with an average atomic mass of 245.6 amu.
Isotope #1 has a mass of 243.2 amu.Isotope #2 has a mass of 244.9 amu.Isotope #3 has a mass of 247.2 amu.Hence, the isotope which is most likely to be the most abundant is isotope #2 that has a mass of 244.9 amu because it is closest in mass to the relative atomic mass (weighted average) of 245.6 amu.
Use the formula: Speed = Distance
Time
3. A snail moves 5 meters across a garden in 2 hours, how fast was the snail?
Answers
A gas is under a pressure of 760torr and occupies volume of 525cm3. If the pressure is doubled, what volume would gas now occupy? Assume the temperature is constant?
Answers
262. 5cm3.
using the Boyles law formula. p1v1=p2v2
P1=760torr.
v1=525cm³
p2=760×2=1520torr.
v2=?
p1v1=p2v2
760×525=1520×v2
v2=[ 760×525]÷1520
v2= 262.5cm³
when the pressure increases, the volume decreases.
What characteristic should an isotope used for dating have?
O A. It should have a half-life much shorter than the age of the object
being dated.
B. It should have a half-life that is close to the age of the object being
dated.
O C. It should have a half-life that changes with time in the object being
dated.
O D. It should have a half-life much longer than the age of the object
being dated.
Answers
Answer:
B
Explanation:
took the test
D. It should have a half-life much longer than the age of the object being dated. When using isotopes for dating purposes, it is important to choose an isotope with a half-life that is significantly longer than the age of the object being dated.
What is the half-life time?The half-life is the time it takes for half of the radioactive isotopes in a sample to decay into stable isotopes. By measuring the ratio of the original radioactive isotope to the decay product, scientists can determine the age of the object.
The majority of the radioactive material would have decayed if the isotope had a half-life substantially less than the age of the artefact, making it challenging to precisely establish the age.
On the other hand, the dating procedure would become more challenging and unpredictable if the isotope in the object being dated (option C) has a half-life that varies over time.
As a result, an isotope with a long half-life offers a stronger basis for precise dating since it enables sufficient radioactive decay to take place, producing quantifiable and trustworthy data.
Learn more about isotope at:
https://brainly.com/question/11680817
#SPJ2
Which step of mitosis involves the nucleus spitting and nuclear membranes forming around each new nucleus??
Answers
Answer:
telophase
Explanation:
All cells must have energy to function. Most of the energy used by the cells in your body is produced by cellular respiration. In cellular respiration, cells use ____________ to release energy stored in __________.
Answers
Answer:
In cellular respiration, cells use oxygen to release energy stored in glucose.
If my answer helped, please mark me as the brainliest!!
Thank You!!
Answer:oxygen and sugars
Explanation:
Rank the following compounds in order of decreasing vapor pressure.
CH3CH2CH2CH2CH3
CH4
CH3CH-CH3CH2CH3
CH3CH2CH2OH
Answers
Decreasing vapour pressure for the following compounds is as follow:
CH4
CH3CH-CH3CH2CH3
CH3CH2CH2CH2CH3
CH3CH2CH2OH
Vapour Pressure of the compound:
The pressure characteristic of a pure compound's vapour at any given temperature when it is in equilibrium with its liquid or solid state is known as the vapour pressure. Compound molecules that bond well with one another will have a low vapor pressure (less inclination to escape to the vapor phase), whereas compounds that connect poorly with one another would have a high vapor pressure. Vapor pressure is a measure of a compound's capacity to bond with itself.
To learn more about the Vapor Pressure:
https://brainly.com/question/4463307
#SPJ1
In lab you measure out 95.12 grams of C4H8O4 for an experiment. How many mol is this? Give your answer to 2 decimal spaces.
Answers
The molar mass of the compound C₄H₈O₄ is 120 g/mol. Then the number of moles of this compound in 95.12 g is 0.79 moles.
What is molar mass ?One mole of every substance contains 6.022 × 10²³ atoms or molecules. This number is called Avogadro number. Thus, one mole of every element contains Avogadro number of atoms.
Similarly one mole of every compound contains Avogadro number of molecules. The mass of one mole of a compound is called its molar mass.
molar mass of C₄H₈O₄ = 120 g/mol (C =12g, O= 16g, H =1 g).
then, number of moles in 95.12 g = given mass/molar mass
no.of moles = 95.12 g /120 g/mol = 0.79 moles.
Therefore, the number of moles of the compound in 95.12 g is 0.79 moles.
Find more on molar mass:
https://brainly.com/question/17010622
#SPJ1
which of the following are examples of single replacement reactions select all that apply.
1) Ca(OH)2(aq) + 2 HCl(aq) ----> CaCl2(aq) + 2 H2O(l)
2)Mg(s) + Zn(NO3)2(aq) ------> Mg(NO3)2(aq) + Zn(s) 3)Na2S(aq) + Cd(NO3)2(aq) ----> 2 NaNO3(aq)
4) K(s) + 2HCl(aq) ----> 2KCl (aq) +H2(g)
Answers
Answer:
4
Explanation:
please mark brainiest
K(s) + 2HCl(aq) ----> 2KCl (aq) +H2(g) is an example of single replacement reactions.
What do you mean single replacement reactions?A single-displacement reaction, also known as single replacement reaction or exchange reaction, is a chemical reaction in which one element is replaced by another in a compound.
Single-replacement reactions always involve two pure elements and one aqueous compound/solution. In the above reaction, the A and C would be pure elements.
A single replacement reaction, sometimes called a single displacement reaction, is a reaction in which one element is substituted for another element in a compound.
Learn more about single replacement reactions:
https://brainly.com/question/13903783
#SPJ2
Calculate the molarity of an aqueous solution that is 22. 3% by mass calcium chloride. You might need to know that the density is 1. 20 g/ml.
Answers
Answer:
Explanation:
To calculate the molarity of the solution, we first need to determine the mass of calcium chloride (CaCl2) in 100 g of the solution.
If the solution is 22.3% calcium chloride by mass, this means that there are 22.3 g of CaCl2 in 100 g of the solution.
We can use the density of the solution to convert 100 g of the solution to its volume:
volume of 100 g of the solution = mass of the solution / density of the solution
volume of 100 g of the solution = 100 g / 1.20 g/mL = 83.3 mL
Now, we can calculate the molarity of the solution using the following formula:
molarity = moles of solute / volume of solution (in L)
The molar mass of CaCl2 is 40.08 + 2(35.45) = 110.98 g/mol. Therefore, the number of moles of CaCl2 in 22.3 g of the solution is:
moles of CaCl2 = 22.3 g / 110.98 g/mol = 0.2008 mol
The volume of the solution containing 22.3 g of CaCl2 is:
volume of solution = 83.3 mL / 1000 mL/L = 0.0833 L
Substituting these values into the formula for molarity, we get:
molarity = 0.2008 mol / 0.0833 L = 2.41 M
Therefore, the molarity of the aqueous solution that is 22.3% by mass calcium chloride is 2.41 M.