Which of these is not a safety decision?
O A. Using a material that will fail in a safe way
OB. Using a material that is sustainable and environmentally friendly
O C. Choosing a material that will show warning before failing
D. Choosing a material that will fail in a predictable way

Answers
Answer:
B is the answer
Explanation:
Answer:the answer was B , he is correcf
Explanation:
Related Questions
All of the orbitals in a given subshell have the same value of the __________ quantum number
Answers
Answer:
magnetic spin
Explanation:
the magnetic spin quantum number i.e + 1/2 or-1/2 ...those are all the orbitals in agiven shell simolarly contains it
Answer:
principal
Explanation:
The quantum numbers that are used in the identification of an electron are n, l, m, and s.
n= principle quantum number
l= azimuthal quantum number
m= magnetic quantum number
s= spin quantum number
All the orbitals in a given subshell have the same value of n(principle quantum number) and l( azimuthal quantum number).
I need ideas, so I have a science project, and I have to decorate a board that goes with my project, I am doing a bottle rocket project with baking soda and vinegar, how should I decorate my board (I can decorate it however I want)
Answers
Answer:
Below :))
Explanation:
I would suggest having a space/rocket theme, which fits your project.
What you do really depends on how much you want to put into it.
If its a black board, you could draw/make rockets to put on the board, and some planets. You could also make/draw some baking soda and vinegar bottles/cans. From what I have done, I have pictures/writing to put on the board. I suggest priting these if you have them, and putting them around the board. You should probably put theses on first, and then draw/make things to put on the board afterward.
Hope this gives you some ideas, if not though, comment down below and I can help more!
What type of reaction will occur if AH is negative and entropy increases?
O spontaneous reaction
O Gibbs free reaction
O exothermic reaction
endothermic reaction
Answers
The type of reaction that will occur if AH is negative and entropy increases is option A which is a spontaneous reaction .
Spontaneous reaction explained.The spontaneity of any chemical reaction depends on the Gibbs free reaction which is related to entropy change and enthalpy change.ΔG= ΔH-TΔS
Where T is temperature change in kelvin.If AH is negative, entropy will increases which is ΔS. Then the ΔG will depend on the temperature change.
If TΔS is larger than ΔH, the reaction will be spontaneous and ΔG will be negative which means the reaction will move forward without any external input.
When a change in entropy increases with the temperature in the system, the reaction will be spontaneous and this will make the Gibbs free energy to be negative.
Learn more about spontaneous reaction below.
https://brainly.com/question/14061406
#SPJ1
moles of each product that would form as a result of the decomposition of aspirin
Answers
The decomposition of aspirin (acetylsalicylic acid,\(C_{9} H_{8} O_{4}\)) can occur through the hydrolysis reaction, resulting in the formation of acetic acid (\(CH_{3} COOH\)) and salicylic acid (\(C_{7} H_{6}O_{3}\)).
The decomposition of aspirin (acetylsalicylic acid, \(C_{9} H_{8} O_{4}\)) can occur through the hydrolysis reaction, resulting in the formation of acetic acid (\(CH_{3} COOH\)) and salicylic acid (\(C_{7} H_{6}O_{3}\)). To determine the moles of each product formed, we need to consider the balanced chemical equation for the reaction:
\(C_{9} H_{8} O_{4} = > C_{7} H_{6}O_{3} +CH_{3} COOH\)
From the equation, we can see that for every 1 mole of aspirin, 1 mole of salicylic acid and 1 mole of acetic acid are produced.
Therefore, the moles of salicylic acid and acetic acid formed will be equal to the number of moles of aspirin that decomposes. If we know the amount of aspirin in moles, we can directly calculate the moles of each product based on stoichiometry.
For more question on aspirin
https://brainly.com/question/25794846
#SPJ8
Part D
After about 30 minutes, observe the substances in the test tubes again. Record your observations. Include any evidence
of a chemical reaction.
A Caution! If you plan to leave your substances unattended, be sure they are in a safe location away from young children
and pets.
BIY X¹ X₂ 10pt
Test tube
1
2
3
4
5
6
AVE
Observation
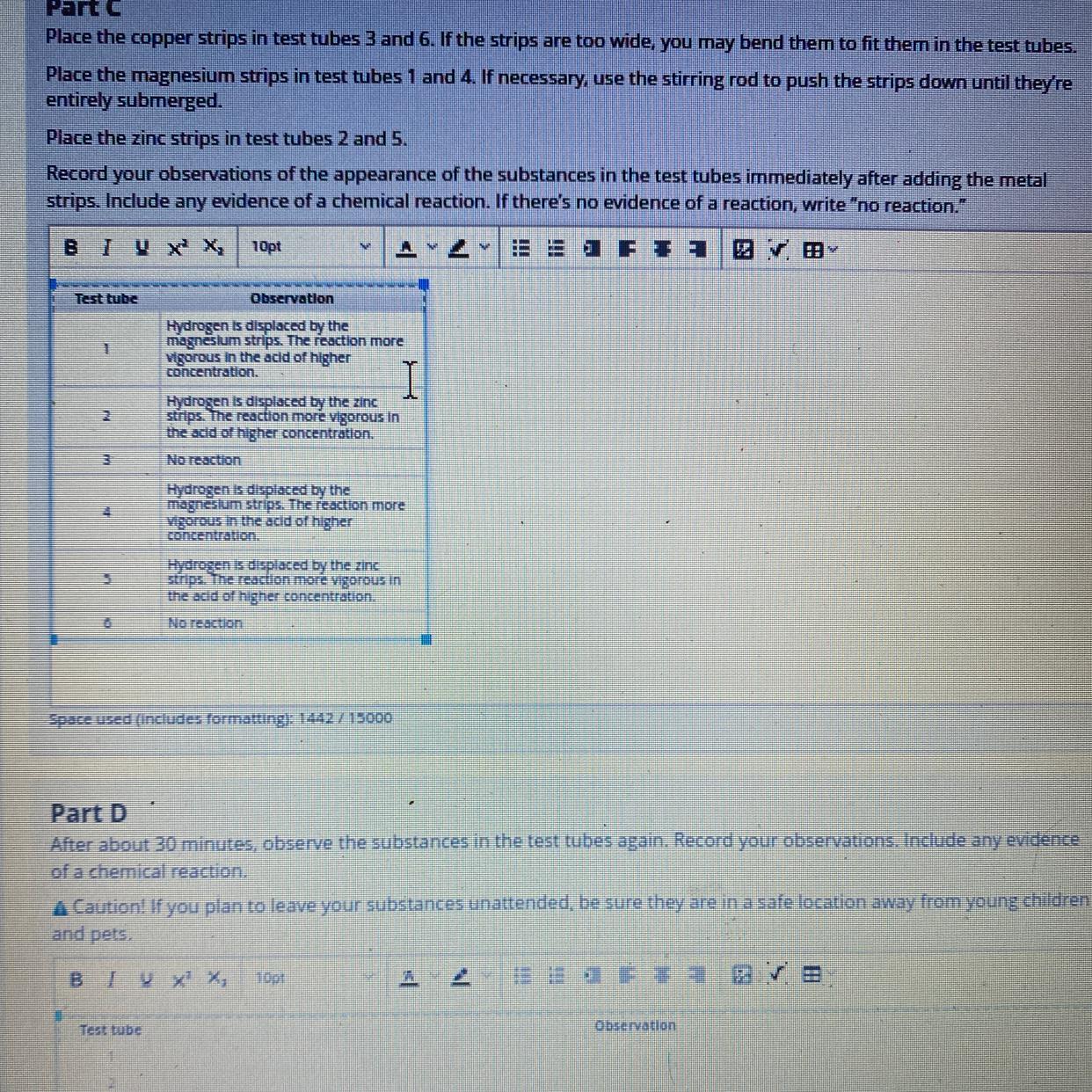
Answers
After about 30 minutes, the color in the test tubes remains same as no more reagents are added and no further chemical change is taking place.
What is a chemical change?Chemical changes are defined as changes which occur when a substance combines with another substance to form a new substance.Alternatively, when a substance breaks down or decomposes to give new substances it is also considered to be a chemical change.
There are several characteristics of chemical changes like change in color, change in state , change in odor and change in composition . During chemical change there is also formation of precipitate an insoluble mass of substance or even evolution of gases.
Learn more about chemical change,here:
https://brainly.com/question/23693316
#SPJ1
A chemist adds 390.0 mL of a 1.5M aluminum chloride (AICI3) solution to a reaction flask. Calculate the mass in grams of aluminum
chloride the chemist has added to the flask. Be sure your answer has the correct number of significant digits.
Answers
Answer:
Calculate the mass in grams.
Answer: Calculate the mass in grams of calcium bromide the chemist has added to the flask. Round your answer to 3 significant digits.
Explanation:
what are the steps to measure the density of a solid?
Answers
Answer:
Look below
Explanation:
The formula for density is:
\(Density=\frac{mass}{volume}\)
Therefore in order to find the density of a solid, you must find the mass and the volume first.
To find the mass, you can use a scale.
To find the volume, you can use the water displacement method. For example, if you fill the water of a graduated cylinder to 10 mL, and then you put a rock inside, and it rises to 15 mL, then the volume (of the rock) is 15mL-10mL=5mL.
A first-order decomposition reaction has a rate constant of 0.00683 yr−1. What is the half-life of the reaction?
Answers
Answer:
101 years
Explanation:
Use the half life equation, where t1/2 is the half life, and k is the rate constant:
t1/2 = 0.693/k
t1/2 = 0.693/0.00683 yr -1
t1/2 = 101 years
Please help I have less than 9 minutes



Answers
And for the multiple choice one I believe it’s D.
Btw I am a middle schooler and college questions are sooo easy especially the medicine ones

Problem 1. What masses of 15% and 20% solutions are needed to prepare 200 g of 17% solution?
Problem 2. What masses of 18% and 5% solutions are needed to prepare 300 g of 7% solution?
Problem 3. 200 g of 15% and 350 g of 20% solutions were mixed. Calculate mass percentage of final solution.
Problem 4. 300 g of 15% solution and 35 g of solute were mixed. Calculate mass percentage of final solution.
Problem 5. 400 g of 25% solution and 150 g of water were mixed. Calculate mass percentage of final solution.
Answers
These are problems related to mixing different solutions to obtain a desired concentration.
According to the given data:Problem 1: 200 g of 17% solution, mix 100 g of 15% solution with 100 g of 20% solution.
Problem 2: 54 g of 18% solution and 246 g of 5% solution are needed.
Problem 3: The mass percentage of the final solution is (20015 + 35020) / (200 + 350) = 18.33%
Problem 4: The mass percentage of the final solution is (30015 + 35) / (300 + 35) = 13.23%
Problem 5: The mass percentage of the final solution is (40025) / (400 + 150) = 21.74%
What is mass?Mass is a fundamental physical property of matter and is defined as the amount of matter that an object contains. It is usually measured in units of grams (g) or kilograms (kg). Mass is related to but different from weight, which is the force exerted on an object by gravity. In chemistry, mass is often used to express the amount of a substance in a sample or reaction.
To know more about mass visit:
https://brainly.com/question/19694949
#SPJ1
what is the product of a reaction 4-acetyl anisole with HI(t°C)?
Answers
Answer:
The reaction of 4-acetyl anisole (also known as p-methoxyacetophenone) with hydroiodic acid (HI) results in the cleavage of the O-methyl group of the anisole ring, leading to the formation of iodobenzene and acetophenone. The balanced equation for this reaction is C9H10O2 + HI → C6H5I + C8H8O.
So, the product of the reaction of 4-acetyl anisole with HI is a mixture of iodobenzene and acetophenone. The specific ratio of these products will depend on the reaction conditions, such as the temperature and concentration of the reactants.
It's worth noting that the reaction between 4-acetyl anisole and HI is an example of a nucleophilic aromatic substitution reaction, which involves the replacement of a leaving group (in this case, the O-methyl group) on an aromatic ring by a nucleophile (the iodide ion from HI).
A biochemist carefully measures the molarity of trypticase in 58.mL of cell growth medium to be 12. μM. Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume of the cell growth medium falls to 16.mL. Calculate the new molarity of trypticase in the cell growth medium.
Answers
Molarity = (moles of solute / volume of solution). Since the moles of solute (trypticase) stays the same, the molarity increases when the volume of solution decreases.
What is the volume ?The volume of a word is an expression of the amount of content in a written piece. It can be measured in terms of the number of words, pages, or characters contained within a text. A text with a high volume will contain more content than a text with a low volume. Plagiarism-free writing is essential for a text to have a high volume, as plagiarized content does not contribute to the overall value of the piece.
The new molarity of trypticase in the cell growth medium would be 48 μM.
Molarity = (moles of solute / volume of solution)
Molarity (original) = (12 μM / 58 mL)
Molarity (new) = moles of solute (original) / volume of solution (new)
Molarity (new) = (12 μM * 58 mL) / 16 mL
Molarity (new) = 48 μM
To learn more about volume
https://brainly.com/question/29796637
#SPJ1
50 points, and I’ll mark as brainliest!!!!!
Tasks are in the picture.

Answers
pH determines the acidic or alkaline a solution is using the pH scale, which has a range of 0 to 14. An alkaline pH is greater than 7, while an acidic pH is less than 7.
Thus, The pH of a solution is defined mathematically as the negative logarithm of the molar concentration of hydrogen ions therein.
NaOH is a strong alkaline, as indicated by a pH testing strip, but in order to determine its exact pH, you must first determine its molarity.
A scale known as pH is used to describe how basic or acidic a water-based solution is. Basic solutions have a higher pH than acidic solutions, which have a lower pH.
Thus, pH determines the acidic or alkaline a solution is using the pH scale, which has a range of 0 to 14. An alkaline pH is greater than 7, while an acidic pH is less than 7.
Learn more about pH, refer to the link:
https://brainly.com/question/15289741
#SPJ1
The pH of HNO₂ is 2.15, pH of NH₄OH is 10.98 and pH of H₂S is 3.76.
pH is defined as the negative logarithm of H⁺ ion concentration.
pH is a measure of how acidic or basic a substance is. In our everyday routine, we encounter and drink many liquids with different pH. Water is a neutral substance. Soda and coffee are often acidic.
The pH is an important property, since it affects how substances interact with one another and with our bodies. In our lakes and oceans, pH determines what creatures are able to survive in the water.
Given,
1. Concentration = 0.1
Ka = 4.5 × 10⁻⁴
\(pH = \frac{1}{2} (pka - log c)\)
pH = 0.5 × ( 3.3 + 1)
= 2.15
2. Concentration = 0.05
Ka = 1.8 × 10⁻⁵
\(pOH = \frac{1}{2} (pkb - log c)\)
pOH = 0.5 × ( 4.74 + 1.3)
= 3.02
pH = 14 - pOH
= 14 - 3.02
= 10.98
3. Concentration = 0.3
Ka = 1 × 10⁻⁷
\(pH = \frac{1}{2} (pka - log c)\)
pH = 0.5 × ( 7 + 0.52)
= 3.76
Learn more about pH, here:
https://brainly.com/question/15289714
#SPJ1
1. The sun warms you. What kind of heat transfer is this an example of
Answers
Answer:
radiation, hope this helps
matching will give brainliest. if you can answer any it help
1. element with atomic number greater than 92.
2. helium nucleus with and atomic number or 2 and a mass of 4.
3. unit for measuring exposure to radiation.
4. changing of one element to another due to alpha or beta decay.
5. caused by the decay of an electron.
6. atoms of the same element with different numbers of neutron.
7. the initial isotope before decay.
a. rem
b. alpha particle
c. beta particle
d. trans uranium element
e. transmutation
f. isotope
g. parent nuclide
Answers
Explanation:
In natural radioactive decay, three common emissions occur. When these emissions were originally observed, scientists were unable to identify them as some already known particles and so named them:
alpha particles ( α )
beta particles (β)
gamma rays (γ)
These particles were named using the first three letters of the Greek alphabet. Some later time, alpha particles were identified as helium-4 nuclei, beta particles were identified as electrons, and gamma rays as a form of electromagnetic radiation like x-rays, except much higher in energy and even more dangerous to living systems.
I NEED THIS DONE TODAY !!!!!!!!Electromagnetic Spectrum Lab Report
Destructions: In this virtual lab, you will use a virtual spectrometer to analyze astronomical
bodies in space. Record your hypothesis and spectrometric recular in the lab report below. You
will submit your completed report to your butructor.
Name and Title:
Include your name, instru
1
and name of lab.
Objectives (1):
In your own words, what is the purpose of this lab?
Hypothesis:
In this section, please include the predictions you developed during your lab activity. These
statements reflect your predicted outcomes for the experiment.
Procedure:
The materials and procedures are listed in your virtual lab. You do not need to repeat them here.
However, you should note if you experienced any errors or other factors that might affect your
outcome. Using your summary questions at the end of your virtual lab activity, please clearly
define the dependent and independent variables of the experiment.
Data:
Record the elements present in each unknown astronomical object. Be sure to indicate "yes" or
"no" for each element.
Hydrogen Helium Lithium Sodiam Carbon
Moon One
Moon Two
Planet One
Planet Two
Nitrogen
Conclusion:
Your conclusion will inchade a summary of the lab results and an interpretation of the results.
Please answer all questions in complete sentences using your own words.
1. Using two to three sentences, summarize what you investigated and observed in this lab
2. Astronomers use a wide variety of technology to explore space and the electromagnetic
spectrum; why do you believe it is essential to use many types of equipment when
studying space?
3. If carbon was the most common element found in the moons and planets, what element is
missing that would make them splat to Earth? Explain why. (Hint: Think about the
carbon cycle)
4.
We know that the electromagnetic spectrum uses wavelengths and frequencies to
determine a lot about outer space. How does it help us find out the make-up of stars?
5. Why might it be useful to determine the elements that a planet or moon is made up of?
PLEASE MAKE SURE YOU ANSWER THE HYPOTHESIS AND PROCEDURE QUESTION!!!!
Answers
Below contains the complete lab report on electromagnetic spectrum
The Lab ReportName: [Your Name]
Title: Electromagnetic Spectrum Lab Report
Instructor: [Instructor's Name]
Objectives:
The purpose of this lab is to analyze the elemental composition of different astronomical bodies using a virtual spectrometer and understand the importance of the electromagnetic spectrum in astronomical research.
Hypothesis:
I predict that the moons and planets will have varying compositions of elements, with hydrogen and helium being more common in gaseous bodies and heavier elements like carbon and nitrogen more common in rocky bodies.
Dependent variable: Presence of elements in astronomical bodies
Independent variable: Astronomical bodies (Moon One, Moon Two, Planet One, Planet Two)
Data:
[Please input your data for each object as per your virtual lab results]
Conclusion:
In this lab, I investigated the elemental composition of four different astronomical bodies using a virtual spectrometer and observed the presence or absence of various elements.
It is essential to use many types of equipment when studying space because different instruments can detect and analyze different aspects of the electromagnetic spectrum, providing a comprehensive understanding of the universe.
To make these moons and planets similar to Earth, oxygen would need to be present as it is a vital component of the carbon cycle and essential for life as we know it.
The electromagnetic spectrum helps us find out the makeup of stars by analyzing the emitted light, which contains information about the elements and their abundance within the star.
Determining the elements that a planet or moon is made up of helps us understand their formation, potential for life, and possible resources for future exploration or colonization.
Read more about Electromagnetic Spectrum Lab Report here:
https://brainly.com/question/30699255
#SPJ1
4. Which is NOT true for ionic compound?
A. usually have high melting and boiling point.
B. characterized by sharing of electrons
C. characterized by transfer of electrons loth Die Hotarw
W
D. forms network of ions.
Answers
(option B) Characterized by sharing of electrons is not true for ionic compounds.
What are ionic compounds?Ionic compounds are chemical compounds formed by the transfer of electrons between a metal and a non-metal. They are composed of positively charged ions (cations) and negatively charged ions (anions) that are held together by electrostatic attractions.
Ionic compounds are formed through the transfer of electrons from one atom to another, resulting in the formation of positively charged cations and negatively charged anions. These oppositely charged ions are held together by strong electrostatic forces of attraction, forming a crystalline lattice structure.
Learn about Ionic compounds here https://brainly.com/question/1332431
#SPJ1
A 275 g sample of a metal requires 10.75 kJ to change its temperature from 21.2 oC to its melting temperature, 327.5 oC. What is the specific heat of this metal
Answers
Answer:
\(c=0.127\ J/g^{\circ} C\)
Explanation:
Given that,
Mass of the sample, m = 275 g
It required 10.75 kJ of heat to change its temperature from 21.2 °C to its melting temperature, 327.5 °C.
We need to find the specific heat of the metal. The heat required by a metal sample is given by :
\(Q=mc\Delta T\)
c is specific heat of the metal
\(c=\dfrac{Q}{m\Delta T}\\\\c=\dfrac{10.75\times 10^3\ J}{275\times (327.5 -21.2)}\\\\=0.127\ J/g^{\circ} C\)
So, the specific heat of metal is \(0.127\ J/g^{\circ} C\).
(a) What mass of lead(II) sulfate would be produced by the action of excess dilute sulfuric acid on 20 g of lead nitrate dissolved in water?
Answers
Mass of lead(II) sulfate would be produced by the action of excess dilute sulfuric acid on 20 g of lead nitrate dissolved in water is 18.19 g.
Lead nitrate react with dilute sulfuric acid : the balanced equation is given below :
Pb(NO₃)₂ + H₂SO₄ -----> PbSO₄ + 2HNO₃
no. of mole = mass / molar mass
moles of Pb(NO₃)₂ = mass / molar mass
= 20 g / 331.2 g/mol
= 0.060 g/mol
1 mole of Pb(NO₃)₂ will produce 1 mole of PbSO₄
0.060 mole of Pb(NO₃)₂ will produced 0.060 mol PbSO₄
therefore, now we can calculate the mass :
no. of moles = mass / molar mass
mass of PbSO₄ = no. of moles × molar mass
= 0.060 × 303.26
= 18.19 g
Thus, Mass of lead(II) sulfate would be produced by the action of excess dilute sulfuric acid on 20 g of lead nitrate dissolved in water is 18.19 g.
To learn more about lead nitrate here
https://brainly.com/question/6851663
#SPJ1
An error during which cellular process would create a gene mutation?
Answers
An error during DNA replication would create a gene mutation.
During DNA replication, the genetic information in a cell is copied to make new DNA molecules. However, mistakes can occur during this process, leading to changes in the DNA sequence, which can result in a mutation. Mutations can also be caused by exposure to environmental factors, such as radiation or chemicals, which can damage the DNA molecule directly or affect the cellular processes involved in DNA replication.
Mutations can have a variety of effects on the organism, ranging from no effect to causing serious health problems or even death. Gene mutations can also be inherited from a parent, which can result in genetic disorders or predisposition to certain diseases. Therefore, it is important to understand the mechanisms of gene mutations and their potential impacts on organisms.
To know more about the Gene mutation, here
https://brainly.com/question/15448555
#SPJ1
Which of the following statements is true about the sense of smell? Men and women smell the same number of odors. The amount of odors one can smell is unknown. Women tend to smell more odors than men. People can smell approximately 100 odors.
Answers
Answer:
the answer is C.) Women tend to smell more odors than men.
Explanation:
The statements is true about the sense of smell is women tend to smell more odors than men.
What is sense?Sense is defined as a biological system utilized by an organism for sensation, which is the process of acquiring information about the environment and responding to stimuli.
Sense organ is defined as a physiological part that is altered by a stimulus in such a way that it causes activation of associated sensory nerve fibers, which transmit specific impulses to the central nervous system, where they are processed as corresponding sensations.
It can also be defined as a portion of the body that allows you to feel the physical features of a scenario.
Ears, eyes, nose, tongue, skin are the five important sense organs of our body.
Thus, the statements is true about the sense of smell is women tend to smell more odors than men.
To learn more about sense, refer to the link below:
https://brainly.com/question/15266656
#SPJ5
Another sample of eggshell reacts completely with 4.0 mL of an HCl(aq) solution of unknown concentration. If the reaction produced 0.095 atm of gas, the concentration of the HCl(aq) solution was at least (A) 0.0020 M (B) 0.050 M (C) 0.50 M (D) 1.0M
Answers
The concentration of the HCl (aq) solution was at least 1.0 M.
What is concentration?The abundance of a constituent divided by the sum of the mixture's volumes is the definition of concentration in chemistry. There are several different categories of mathematical description: mass concentration, molar concentration, number concentration, and volume concentration
P = 0.095atm(corresponds to 0.20g of CaCO3
so,moles of CaCO3 =0.20g/100gmol⁻¹=0.002moles
moles of HCl =(molesₓCaCO3ₓ2)=0.002ₓ2
M=0.004/4ₓ100
Concentration of HCl=1M
For more information about concentration please visit:
https://brainly.com/question/10725862
#SPJ4
Percent Change of pH = 100% x ( pH at 5 drops - pH at 0 drops ) / ( pH at 0 drops )
Answers
The formula you provided calculates the percent change in pH based on the difference between the pH at 5 drops and the pH at 0 drops. Here's how you can use the formula:
1. Determine the pH at 5 drops and the pH at 0 drops.
Let's say the pH at 5 drops is 4 and the pH at 0 drops is 7.
2. Plug the values into the formula:
Percent Change of pH = 100% × (pH at 5 drops - pH at 0 drops) / pH at 0 drops
Percent Change of pH = 100% × (4 - 7) / 7
3. Calculate the numerator:
4 - 7 = -3
4. Calculate the denominator:
Percent Change of pH = 100% × (-3) / 7
5. Calculate the percent change:
Percent Change of pH = -300% / 7
Therefore, the percent change in pH from 0 drops to 5 drops is approximately -42.86%.
If you add one packet of sugar to your tea, and all of the sugar crystals dissolve without
settling down on the bottom of your glass, this solution is
Answers
Answer:
b
Explanation:
sugar crystals are amzing
What kinds of things on earth are made of cells?
Answers
Answer:
All living things, large or small, plant or animal, are made up of cells. Most living things are made up of one cell and they are called unicellular organisms. Many other living things are made up of a large number of cells that form a larger plant or animal. These living things are known as multicellular organisms.
Explanation:
compound composed of cations and anions the forces of attraction that bind oppositely charged ions together lowest whole-number ratio of ions in an ionic compound a depiction of the valence electrons as dots around the symbol for an element a mixture of two or more elements, at least one of which is a metal the attraction of the free-floating valence electrons for the positively charged metal ions ions of the halogen atoms the electron(s) in the highest occupied energy level of an atom the number of ions of opposite charge that surround each ion in a crystal Atoms in a compound tend to have the electron configuration of a noble gas. Shows the kinds and numbers of atoms in the smallest representative unit of a substance a. halide ions b. alloy c. octet rule d. formula unit e. electron dot structure f. ionic compound g. ionic bonds h. coordination number i. chemical formula j. valence electrons k. metallic bond
Answers
Answer:
compound composed of cations and anions the forces of attraction that bind oppositely charged ions together - ionic compound
lowest whole-number ratio of ions in an ionic compound - formula unit
a depiction of the valence electrons as dots around the symbol for an element - electron dot structure
a mixture of two or more elements, at least one of which is a metal - alloy
the attraction of the free-floating valence electrons for the positively charged metal - metallic bond
ions of the halogen atoms - halide ions
the electron(s) in the highest occupied energy level of an atom - valence electrons
the number of ions of opposite charge that surround each ion in a crystal - coordination number
Atoms in a compound tend to have the electron configuration of a noble gas - octet rule
Shows the kinds and numbers of atoms in the smallest representative unit - chemical formula
Explanation:
An ionic compound is composed of oppositely charged ions held together in a crystal lattice. The lowest whole-number ratio of ions in an ionic compound is known as the formula unit. The number of ions of opposite charge that surround each ion in a crystal is called the coordination number. Ions of the halogen atoms are negatively charged and are called halide ions
Elements are composed of atoms, the electron(s) in the highest occupied energy level of an atom are called valence electrons. In metals, the attraction of the free-floating valence electrons for the positively charged metal is called the metallic bond. For atoms of elements, a depiction of the valence electrons as dots around the symbol for an element is called the electron dot structure or Lewis structure.
A combination of elements may be a compound or an alloy. An alloy usually contains at least one metal. For compounds, the chemical formula Shows the kinds and numbers of atoms in the smallest representative unit. Atoms form compounds because atoms in a compound tend to have the electron configuration of a noble gas this is the octet rule.
The correct match of the given statement are 1 - f. ionic compound, 2- d. formula unit, 3 - e. electron dot structure, 4- b. alloy, 5- k. metallic bond, 6- a. halide ions, 7- j. valence electrons, 8- h. coordination number, 9 - c. octet rule, 10- i. chemical formula.
Ionic compounds are made up of oppositely charged ions that are kept together in a crystal lattice. The formula unit is the lowest whole-number ratio of ions in an ionic compound.
The coordination number is the number of ions with opposite charges that surround each ion in a crystal. Halide ions are halogen atom ions that are negatively charged.
Elements are made up of atoms, and the electron(s) in the atom's highest occupied energy level are known as valence electrons. The metallic bond refers to the attraction of free-floating valence electrons to positively charged metals in metals.
The electron dot structure or Lewis structure is a representation of the valence electrons as dots around the symbol for an element for atoms.
A compound or an alloy is a mixture of components. A metal is frequently present in an alloy.
The chemical formula is used for compounds. The types and numbers of atoms in the smallest representative unit are displayed.
The octet rule states that atoms in a compound tend to have the electron configuration of a noble gas.
Therefore, the correct matches are: 1 - f. ionic compound, 2- d. formula unit, 3 - e. electron dot structure, 4- b. alloy, 5- k. metallic bond, 6- a. halide ions, 7- j. valence electrons, 8- h. coordination number, 9 - c. octet rule, 10- i. chemical formula.
Learn more about ionic compound, here:
https://brainly.com/question/30420333
#SPJ6
The given question is incomplete, so the most probable complete question is,
Match the correct statement with their appropriate terms:
Compound composed of cations and anions, the forces of attraction that bind oppositely charged ions together.Lowest whole-number ratio of ions in an ionic compound.A depiction of the valence electrons as dots around the symbol for an element.A mixture of two or more elements, at least one of which is a metal.The attraction of the free-floating valence electrons for the positively charged metal.Ions of the halogen atoms.The electron(s) in the highest occupied energy level of an atom.The number of ions of opposite charge that surround each ion in a crystal.Atoms in a compound tend to have the electron configuration of a noble gas.Shows the kinds and numbers of atoms in the smallest representative unit.during a baseball game the sound of the bat hitting the ball
Answers
During a baseball game, the sound of the bat hitting the ball can be heard in most parts of the stadium. That sound is weaker at greater distances. The cause of this phenomenon is inverse-square law.
What is sound?Sound is a physical disturbance from an equilibrium condition that travels via an elastoplastic medium. A completely subjective definition of sound, as perceived by the ear, is also viable, but it is not especially informative and is overly limited, because it is important to speak of noises which can be heard by the auditory system, such as those produced by dog whistles or sonar technology.
The inverse-square law, that states that the strength of a sound wave is inversely related to the square of the distance to the source, is the cause of this phenomena. The strength of such sound wave reduces as the distance to the source rises.
Therefore, the cause of this phenomenon is inverse-square law.
To learn more about sound, here:
https://brainly.com/question/733324
#SPJ1
38 mL of a gas was collected at 581 mm Hg and 25 degrees Celsius. What would the volume be at standard pressure (560 mm Hg) if the temperature remained constant?
Answers
Khpo4 is mono basic salt but when I write potassium phosphate mono basic then I get kh2po4
Answers
It is a salt formed from the potassium cation (K+) and the phosphate anion (H2PO4-). This salt is considered monobasic because it contains one replaceable hydrogen ion (H+).
KH2PO4 is actually potassium dihydrogen phosphate, not potassium phosphate monobasic. Potassium phosphate monobasic, or monopotassium phosphate, is correctly represented by the chemical formula KH2PO4. It is a salt formed from the potassium cation (K+) and the phosphate anion (H2PO4-). This salt is considered monobasic because it contains one replaceable hydrogen ion (H+).
On the other hand, potassium dihydrogen phosphate, or K2HPO4, is a different compound. It is formed from the potassium cation (K+) and the hydrogen phosphate anion (HPO4^2-). This compound is considered dibasic because it contains two replaceable hydrogen ions (H+).
Therefore, KH2PO4 is correctly identified as potassium phosphate monobasic, while K2HPO4 is potassium dihydrogen phosphate.
For more question on cation
https://brainly.com/question/14309645
#SPJ8
An unknown liquid has a heat of vaporization of 5.48 kJ/mole. If the vapor pressure of this liquid at -170 degrees C is 117 torr, what is the normal boiling point of this liquid in degrees C? HINT: Normal boiling point occurs when the vapor pressure of the liquid is the same as atmospheric pressure (1 atm or 760 mm Hg).
Answers
The normal boiling point of the unknown liquid is 57.4°C.
The normal boiling point occurs when the vapor pressure of the liquid is equal to the atmospheric pressure. At normal boiling point, the temperature of the liquid is called the boiling point.
Using the Clausius-Clapeyron equation:
ln(P₂/P₁) = -(ΔHvap/R) * (1/T₂ - 1/T₁)
where P₁ is the vapor pressure at the given temperature T₁, P₂ is the vapor pressure at the boiling point temperature T₂, ΔHvap is the heat of vaporization, R is the gas constant.
At -170°C, the vapor pressure of the liquid is given as P₁ = 117 torr. At normal boiling point, the vapor pressure of the liquid is P₂ = 760 torr.
Converting all units to SI units, we have:
P₁ = 15.47 Pa
P₂ = 101325 Pa
ΔHvap = 5480 J/mol
R = 8.314 J/(mol*K)
Plugging in the values, we get:
㏑(101325/15.47) = -(5480/8.314) * (1/T₂ - 1/103.15)
Solving for T₂, the boiling point is found to be:
T₂ = 57.4°C
As a result, the unknown liquid's usual boiling point is 57.4°C.
To know more about the Pressure, here
https://brainly.com/question/14748171
#SPJ1